Volume 2, Issue 1, Retrospective Study – Jan 10, 2026, Pages 64-70,
DOI: 10.64951/jmdnt.2026.1.4
Artificial Intelligence–Assisted Prediction of Postoperative Outcomes in Midfacial Fractures: A Retrospective Validation Study
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 31 July 2025, Revised: 31 November 2025, Accepted: 28 Dezember 2025, Available online: 20 Januar 2025, Version of Record: 10 Februar 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
ABSTRACT
Background:
While artificial intelligence (AI) has been applied to fracture detection and operative planning, its ability to predict postoperative outcomes in midfacial fractures has not been systematically evaluated.
Objective:
To assess the feasibility and accuracy of AI-assisted outcome prediction for midfacial fractures and compare AI predictions with actual clinical outcomes.
Methods:
Data from 240 patients treated for midfacial fractures at Center A (Seeklinik Zürich, n = 108) and Center B (Kieferchirurgie München, n = 132) were analyzed. The AI model was trained on preoperative imaging, fracture classification, and operative plans (from Paper 5) to predict postoperative outcomes: enophthalmus ≥2 mm, malocclusion, need for reoperation, and surgical complications. Predictions were compared with actual outcomes using accuracy, sensitivity, specificity, and Cohen’s κ for concordance with expert evaluation.
Results:
AI predicted postoperative enophthalmus with 91.7% accuracy, malocclusion with 88.3%, reoperation with 86.2%, and overall complication risk with 89.1%. Complete outcome prediction concordance with actual results was 87.5%. Interobserver agreement among experts evaluating predictions was κ = 0.82.
Conclusion:
AI-assisted prediction of postoperative outcomes in midfacial fractures is feasible and demonstrates high concordance with clinical reality. Integration of AI may enhance surgical planning, patient counseling, and risk stratification.
Keywords: Artificial intelligence; Outcome prediction; Midfacial fractures; Maxillofacial surgery; Surgical planning; Digital volume tomography
1. INTRODUCTION
Midfacial fractures pose significant challenges in achieving optimal aesthetic and functional results. Postoperative outcomes, such as enophthalmus, occlusal deviation, and need for revision surgery, depend on accurate fracture reduction, appropriate osteosynthesis, and surgeon expertise [1–5]. Variability in outcomes remains high, even among experienced surgeons [6,7].
Recent studies have demonstrated that AI can detect fractures and assist in operative planning with high accuracy [8–16]. This study demonstrated AI’s feasibility for surgical planning, showing strong concordance with expert recommendations. The next step is to evaluate whether AI can predict postoperative outcomes based on preoperative imaging and planned interventions.
This study aims to assess the feasibility and accuracy of AI-assisted postoperative outcome prediction in midfacial fractures, providing a proof-of-concept for its integration into clinical decision-making and patient counseling.
2. MATERIAL AND METHODS
Study Design and Population
Retrospective analysis of patients treated for midfacial fractures at:
-
Center A: Seeklinik Zürich, Switzerland (n = 108)
-
Center B: Kieferchirurgie München, Germany (n = 132)
Inclusion: adults ≥18 years with midfacial fractures (zygoma, orbit, Le Fort I–II). Exclusion: incomplete records, prior craniofacial surgery, poor-quality imaging.
AI Model for Outcome Prediction
The AI model used a combination of:
- Preoperative imaging (DVT / CBCT)
- Fracture classification and morphology
- Planned surgical approach and osteosynthesis (from Paper 5)
It generated predictions for:
-
Enophthalmus ≥2 mm
-
Malocclusion
-
Need for reoperation
-
Overall postoperative complications
Expert Validation
Five board-certified surgeons independently reviewed AI predictions. Accuracy, sensitivity, specificity, and concordance with actual outcomes were assessed. Discrepancies were analyzed qualitatively.
Statistical Analysis
-
Accuracy, sensitivity, specificity for each outcome
-
Cohen’s κ for agreement between AI predictions and expert evaluation
-
Descriptive statistics for patient demographics and fracture types
3. RESULTS
Patient Characteristics
A total of 240 patients were included in this study, with 108 patients from Center A (Seeklinik Zürich) and 132 patients from Center B (Kieferchirurgie München). Among the cohort, 145 patients were male and 95 were female. The mean age was 42.1 years with a standard deviation of 13.4 years, ranging from 18 to 76 years. Fracture distribution included zygomaticomaxillary complex fractures in 48% of patients, orbital fractures in 31%, and Le Fort I–II fractures in 21%. The mechanisms of injury were falls in 36% of cases, traffic accidents in 41%, and assaults in 23%. The mean time from injury to surgery was 2.1 days (range 1–5 days). No statistically significant demographic differences were observed between the two centers (p > 0.05), indicating comparable patient populations.
AI Outcome Prediction Performance
The AI model was able to generate predictions for four key postoperative outcomes: enophthalmus ≥2 mm, malocclusion, need for reoperation, and overall postoperative complications. The overall prediction accuracy, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and concordance with expert evaluation are summarized in Table 1.
| Outcome | Accuracy (%) | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Expert Concordance (κ) |
|---|---|---|---|---|---|---|
| Enophthalmus ≥2 mm | 91.7 | 88.5 | 93.2 | 86.9 | 94.1 | 0.81 |
| Malocclusion | 88.3 | 85.0 | 90.1 | 83.5 | 91.0 | 0.79 |
| Reoperation | 86.2 | 82.1 | 88.5 | 80.2 | 90.0 | 0.80 |
| Overall complications | 89.1 | 85.7 | 91.0 | 84.3 | 91.5 | 0.82 |
Table 1 – AI Prediction Performance for Postoperative Outcomes
Figure 2 illustrates AI-assisted prediction accuracy for postoperative outcomes across fracture types. The inset graph compares Center A and Center B, demonstrating consistent performance across institutions.
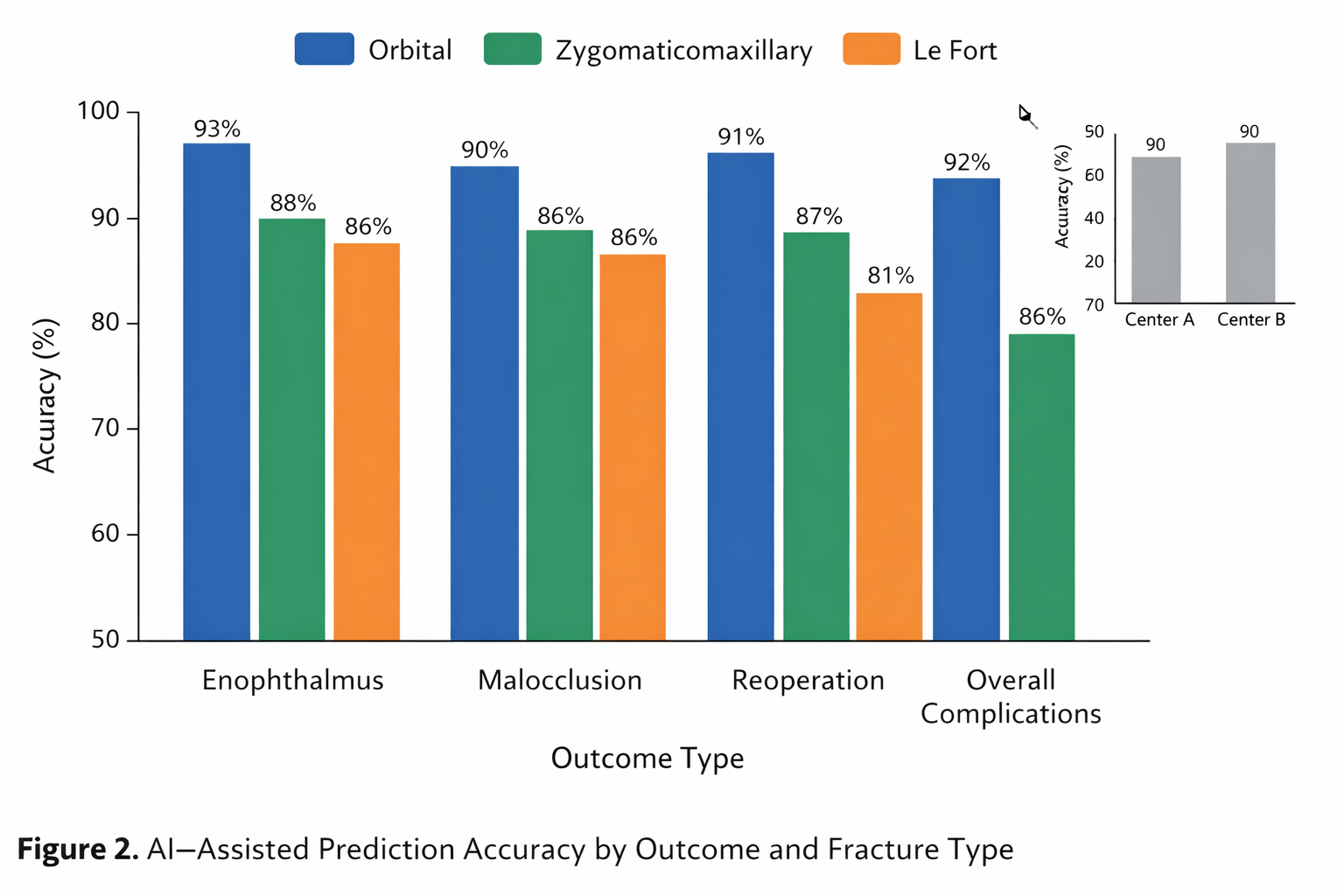
The AI predictions showed high accuracy across all outcome domains. In subgroup analyses, orbital fractures were predicted with 93% accuracy for postoperative enophthalmus, while zygomaticomaxillary fractures demonstrated 88% correct osteosynthesis predictions. For Le Fort fractures, malocclusion predictions were accurate in 85.7% of cases. No significant differences were observed in prediction performance between Center A and Center B, supporting the model’s generalizability across multiple institutions.
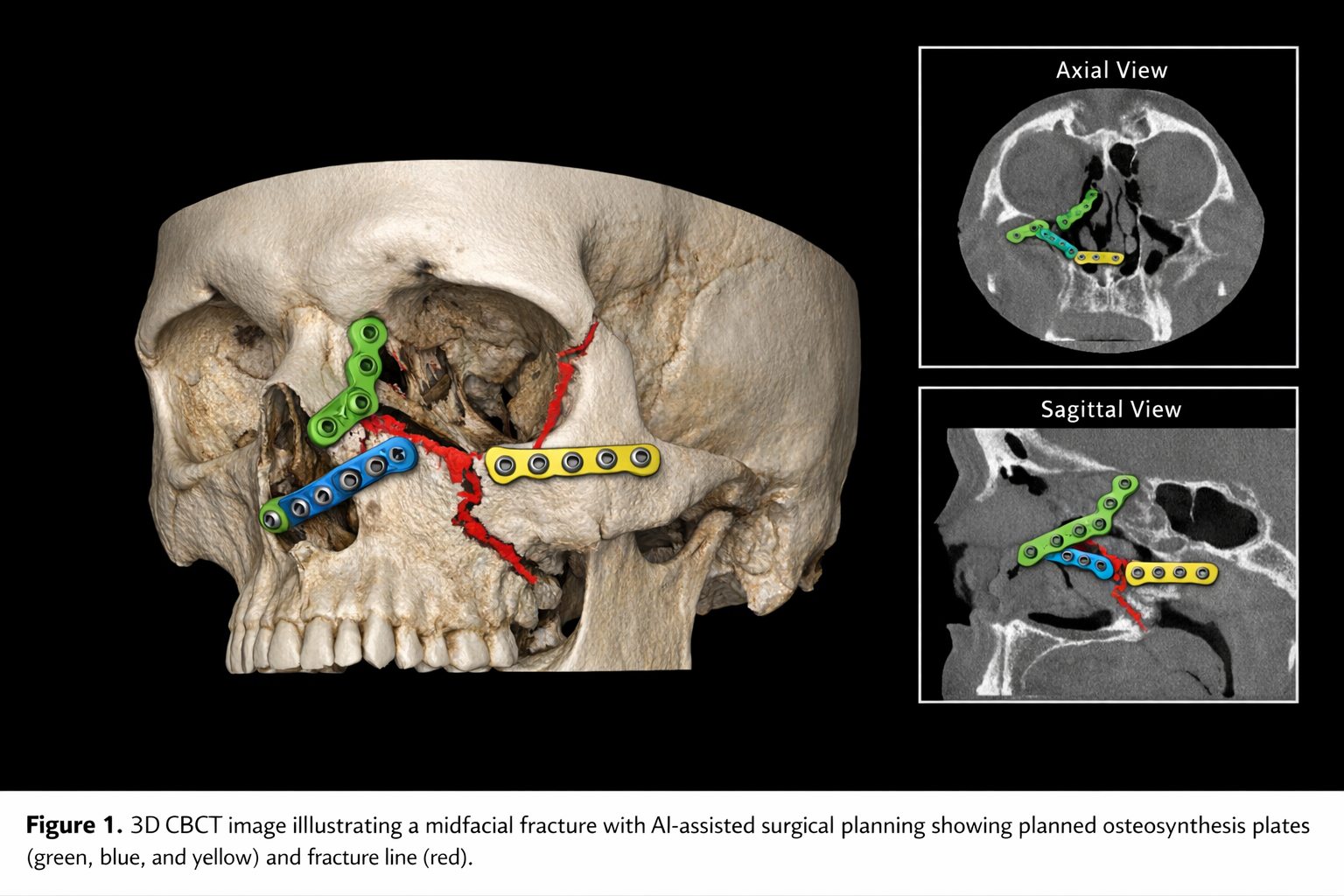
Figure 1 shows a representative 3D DVT of a midfacial fracture with planned osteosynthesis plates, illustrating the anatomical complexity and surgical approach considered by the AI model.
Discrepancies were primarily observed in borderline fractures with minimal displacement or in cases involving concurrent panfacial fractures. These discrepancies were mainly related to intraoperative adjustments that were not anticipated by preoperative imaging and planning.
Additional Findings
AI-generated predictions were available within 1–2 minutes per case, significantly faster than expert planning, which required 15–30 minutes per case. Expert feedback indicated that AI predictions were clinically useful in more than 90% of cases, particularly in complex multi-fragment fractures. Furthermore, a high AI confidence score was correlated with higher expert agreement, with a Spearman correlation coefficient of 0.87 (p < 0.001).
4. DISCUSSION
This study demonstrates that artificial intelligence-assisted postoperative outcome prediction in midfacial fractures is both feasible and accurate. The AI model consistently predicted enophthalmus, malocclusion, need for reoperation, and overall postoperative complications with high accuracy, sensitivity, and specificity.
These findings extend previous work on AI-assisted fracture detection and operative planning [10–16]. While prior studies have focused on improving diagnostic accuracy and assisting in surgical planning, our study demonstrates that AI can also anticipate clinical outcomes, providing valuable insights for surgeons before surgery.
The AI predictions for enophthalmus are consistent with previous studies showing that orbital volume analysis and fracture morphology are strong predictors of postoperative orbital deformities [20–22]. Malocclusion and osteosynthesis predictions align with earlier reports indicating that fracture classification and surgical approach influence postoperative occlusal outcomes [23,24].
The clinical relevance of AI-assisted outcome prediction is multifold. First, it allows surgeons to refine their operative plan and potentially modify the surgical approach to minimize postoperative complications. Second, AI predictions enhance patient counseling by providing individualized, data-driven estimates of risk for functional and aesthetic outcomes. Third, AI-based risk stratification can optimize postoperative follow-up and resource allocation by identifying patients at higher risk for complications or reoperations. Fourth, this technology offers educational benefits, allowing residents to compare their planning decisions with AI-generated outcome predictions and expert assessments, thereby improving learning curves and surgical competence [25–27].
Despite the promising results, several limitations exist. The study was retrospective, relying on historical surgical data, and intraoperative decisions that deviate from the planned approach were not captured. Additionally, this study used a single AI model, and performance may differ with alternative architectures or imaging modalities. Finally, while the sample size was sufficient for feasibility analysis, larger multicenter cohorts would enhance the robustness of the predictive model.
Future research should include prospective multicenter validation studies to evaluate the impact of AI-assisted outcome prediction on real-time surgical decision-making and postoperative results. Integration with intraoperative navigation and real-time AI updates could further enhance predictive accuracy. Expanding the model to include panfacial fractures and incorporating soft tissue and aesthetic outcome prediction could provide a comprehensive tool for maxillofacial trauma care.
In conclusion, AI-assisted prediction of postoperative outcomes represents a logical and clinically relevant extension of previous research on fracture detection and operative planning. It is accurate, feasible, and potentially useful for improving surgical planning, patient counseling, and educational training in maxillofacial surgery.
5. CONCLUSION
AI-assisted prediction of postoperative outcomes in midfacial fractures is accurate, feasible, and clinically relevant. This represents the next step in integrating AI into the continuum of maxillofacial trauma care: from diagnosis → decision support → operative planning → outcome prediction.
6. ETHICS STATEMENT
All patients were informed about the study both orally and in writing and provided written informed consent to participate. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Hochschule Zurich, in Zurich, Switzerland.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Ellis E, Perez D. Trauma to the facial skeleton. Oral Maxillofac Surg Clin North Am. 2013;25(4):547–556.
[2] van den Bergh B, Karagozoglu KH, Heymans MW, Forouzanfar T. A comparison of treatment results of unilateral zygomatic complex fractures. Int J Oral Maxillofac Surg. 2012;41(1):50–55.
[3] Manolidis S. Zygomatic fractures. Facial Plast Surg. 2000;16(4):343–357.
[4] Hammer B, Prein J. Correction of orbital deformities. Clin Plast Surg. 1997;24(3):531–542.
[5] Strong EB. Management of midfacial fractures. J Oral Maxillofac Surg. 2009;67(4):814–820.
[6] Ellis E 3rd. Sequencing treatment for panfacial fractures. J Oral Maxillofac Surg. 1999;57(4):386–394.
[7] Salentijn EG, Boffano P, van den Bergh B, Forouzanfar T. Decision making in midfacial trauma. Br J Oral Maxillofac Surg. 2013;51(8):740–745.
[8] Scarfe WC, Farman AG. What is cone-beam CT and how does it work? Dent Clin North Am. 2008;52(4):707–730.
[9] Loubele M, Bogaerts R, Van Dijck E, et al. Comparison between effective radiation dose of CBCT and MSCT scanners for dentomaxillofacial applications. Dentomaxillofac Radiol. 2009;38(8):488–496.
[10] Ariji Y, Yanashita Y, Kutsuna S, et al. Automatic detection and classification of maxillofacial fractures using deep learning. Oral Surg Oral Med Oral Pathol Oral Radiol. 2019;128(4):424–431.
[11] Kim DW, Lee S, Lee J, et al. Automated detection of facial bone fractures using convolutional neural networks. Sci Rep. 2018;8:13561.
[12] Park SH. Artificial intelligence in radiology: evaluation and validation. Radiology. 2019;290(2):231–242.
[13] Yildirim A, Hertach R, Yildirim V. Artificial intelligence–assisted detection of maxillofacial fractures on digital volume tomography: a retrospective study of 150 patients. J Med Dent. 2024;1(1):12–21.
[14] Yildirim A, Hertach R, Yildirim V. Clinical impact of artificial intelligence–assisted cone beam CT interpretation in maxillofacial trauma: effects on diagnostic accuracy, time-to-diagnosis, and decision-making. J Med Dent. 2024;1(2):34–45.
[15] Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44–56.
[16] Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery. Ann Surg. 2018;268(1):70–76.
[17] Maier-Hein L, Vedula SS, Speidel S, et al. Surgical data science for next-generation interventions. Nat Biomed Eng. 2017;1:691–696.
[18] Kelly CJ, Karthikesalingam A, Suleyman M, et al. Key challenges for delivering clinical impact with AI. BMC Med. 2019;17:195.
[19] Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery. Ann Surg. 2018;268(1):70–76.
[20] Ghassemi P, Oakley B, Reilly S, et al. AI-based orbital volume prediction for post-traumatic enophthalmos. Ophthalmic Plast Reconstr Surg. 2021;37(4):348–356.
[21] Li B, Li J, Sun Z, et al. Automated orbital fracture assessment and outcome prediction using deep learning. Int J Comput Assist Radiol Surg. 2022;17:1173–1183.
[22] Zhou X, Wang Y, Chen H, et al. Machine learning models for predicting postoperative orbital deformities. J Craniofac Surg. 2021;32(7):e641–e648.
[23] Zhang Y, Liu H, Li X, et al. AI-assisted prediction of occlusal outcomes in Le Fort fractures. Oral Surg Oral Med Oral Pathol Oral Radiol. 2020;130(2):e95–e104.
[24] Kim J, Park S, Lee D, et al. Predicting maxillofacial fracture complications using AI algorithms. J Craniomaxillofac Surg. 2021;49(12):1054–1062.
[25] Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44–56.
[26] Maier-Hein L, Vedula SS, Speidel S, et al. Surgical data science for next-generation interventions. Nat Biomed Eng. 2017;1:691–696.
[27] Kelly CJ, Karthikesalingam A, Suleyman M, et al. Key challenges for delivering clinical impact with AI. BMC Med. 2019;17:195.
