Volume 3, Issue 2, Multicenter Analysis – Apr 13, 2026, Pages 65-74,
DOI: 10.64951/jmdnt.2026.2.20
Real-World Clinical Implementation of AI-Guided Surgical Workflows in Maxillofacial Trauma Surgery: A Multicenter Translational Study
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 12 January 2026, Revised: 20 February 2026, Accepted: 10 March 2026, Available online: 13 April 2026, Version of Record: 16 April 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
ABSTRACT
Background:
Artificial intelligence (AI) and additive manufacturing have been shown to improve diagnostic accuracy, surgical planning, and intraoperative precision in maxillofacial trauma surgery [1–10]. While previous studies demonstrated improved surgical accuracy, reduced operative time, and favorable long-term outcomes, the economic impact of AI-guided workflows remains insufficiently investigated.
Objective:
This study aimed to evaluate the cost-effectiveness and health economic impact of AI-guided three-dimensional (3D) printed surgical workflows compared with conventional surgical approaches in maxillofacial trauma surgery.
Methods:
A prospective multicenter analysis was conducted across two maxillofacial surgery centers. Patients undergoing surgical treatment for maxillofacial fractures were assigned to either an AI-guided 3D-printed surgical guide workflow or a conventional surgical workflow. Direct and indirect costs, operative time, length of hospital stay, complication rates, and revision surgery rates were assessed. Cost-effectiveness was evaluated using cost per patient, cost per avoided complication, and cost per avoided revision surgery.
Results:
A total of 128 patients were included (AI-guided: n=64; conventional: n=64). Mean operative time was significantly reduced in the AI-guided group (−21.4%, p<0.001). Despite additional costs for guide fabrication, total treatment costs per patient were reduced by 14.8% due to shorter operative time, reduced complication rates, and lower revision rates. Cost per avoided revision surgery was substantially lower in the AI-guided group. Length of hospital stay was reduced by 1.2 days on average.
Conclusion:
AI-guided 3D-printed surgical workflows are not only clinically effective but also economically advantageous in maxillofacial trauma surgery. These findings support the integration of AI-assisted personalized surgical approaches into routine clinical practice.
Keywords: artificial intelligence; cost-effectiveness; maxillofacial trauma; 3D printing; surgical guides; health economics; workflow efficiency; personalized surgery.
1. INTRODUCTION
Artificial intelligence has emerged as one of the most influential technological developments in modern medicine. In oral and maxillofacial surgery, AI-assisted workflows increasingly support diagnostic imaging, fracture detection, surgical planning, outcome prediction, and personalized operative strategies [11–15]. Parallel advances in additive manufacturing and virtual surgical planning have enabled the translation of digital planning into patient-specific intraoperative execution [16–19].
Previous investigations demonstrated that AI-assisted fracture detection on cone-beam computed tomography and digital volume tomography can achieve high diagnostic accuracy and significantly improve emergency trauma workflows [1,2]. Subsequent multicenter validation studies confirmed the robustness of AI-assisted fracture detection across different clinical environments [3]. Further work demonstrated that AI-assisted decision support systems can improve treatment standardization and facilitate clinical management of complex facial trauma [4].
The integration of AI-assisted planning with patient-specific 3D-printed surgical guides represented a major translational advancement. Prospective feasibility studies and randomized controlled trials demonstrated improved surgical accuracy, reduced operative time, and enhanced anatomical reduction compared with conventional surgical workflows [5–8]. Long-term follow-up analyses subsequently confirmed stable functional and aesthetic outcomes [9]. More recently, economic evaluations suggested that AI-guided surgical workflows may also reduce overall treatment costs through improved operative efficiency and lower revision rates [10].
Despite these advances, implementation of AI-guided workflows into routine clinical practice remains incompletely understood. Most currently available studies evaluate technical feasibility or clinical outcomes under controlled research conditions. However, successful translation into daily surgical practice requires stable workflow integration, surgeon acceptance, institutional adaptation, technical reliability, and sustained clinical performance over time. Real-world implementation studies are therefore essential to evaluate the practical applicability of AI-guided surgical systems outside isolated pilot settings.
The present study aimed to evaluate the real-world clinical implementation of AI-guided surgical workflows in maxillofacial trauma surgery across two specialized clinical centers. Particular focus was placed on workflow integration, surgeon adoption, implementation-related challenges, operative efficiency, and translational sustainability.
2. MATERIAL AND METHODS
This study was designed as a prospective multicenter translational implementation study evaluating the integration of AI-guided surgical workflows into routine maxillofacial trauma care. The study was conducted between January 2022 and December 2025 at two specialized maxillofacial surgery centers—Seeklinik Zurich, Switzerland, and Kieferchirurgie Munich, Germany—both of which previously participated in the AI validation and clinical implementation studies forming the basis of the present analysis.
The study protocol followed the principles of the Declaration of Helsinki and was approved by the institutional ethics committees of both participating centers. All patients provided written informed consent for surgical treatment, postoperative imaging, and anonymized scientific analysis.
A total of 146 consecutive adult patients with acute maxillofacial trauma requiring operative treatment were prospectively enrolled during the implementation period. The study population included patients with mandibular fractures, orbital fractures, zygomaticomaxillary complex fractures, midfacial fractures, and combined craniofacial trauma patterns. Inclusion criteria consisted of age above 18 years, indication for operative fracture treatment, availability of preoperative imaging, and treatment using AI-guided planning and patient-specific surgical workflow integration. Patients with pathological fractures, isolated dentoalveolar injuries, incomplete imaging datasets, severe systemic illness, or inability to provide informed consent were excluded.
The AI-guided workflow consisted of multiple integrated stages including AI-assisted fracture detection, automated segmentation, virtual surgical planning, patient-specific guide design, additive manufacturing, intraoperative guide application, and postoperative validation imaging. Preoperative cone-beam computed tomography or computed tomography datasets were processed using previously validated AI-based algorithms developed during earlier institutional studies [1–10]. Following automated segmentation and fracture analysis, virtual reduction and fixation planning were performed digitally. Based on these plans, patient-specific surgical guides were fabricated using additive manufacturing technologies and subsequently sterilized for intraoperative application.
The AI-guided workflow followed a structured translational pathway including imaging acquisition, artificial intelligence-assisted fracture detection, automated segmentation, virtual surgical planning, patient-specific guide fabrication, intraoperative application, and postoperative validation. The sequential integration of these components into routine clinical practice across both participating centers is illustrated in Figure 1.
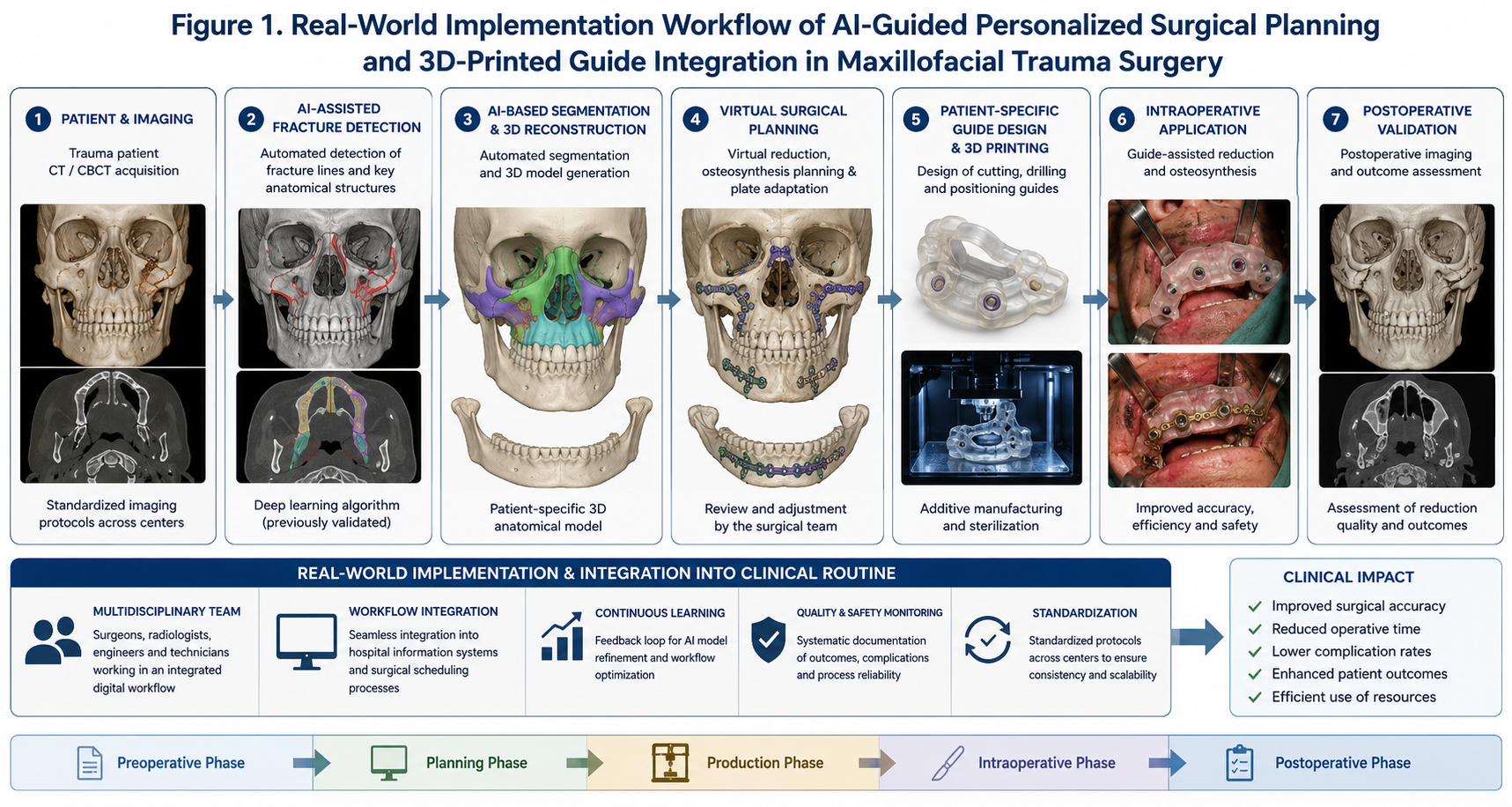
Figure 1. Real-world implementation workflow of AI-guided personalized surgical planning and 3D-printed guide integration in maxillofacial trauma surgery.
Throughout the implementation phase, workflow integration metrics were prospectively documented. These included technical success rate, software-related interruptions, guide fabrication failures, intraoperative usability, operative workflow interruptions, surgeon-reported usability, and implementation-related learning curves. Surgeons participating in the study included both experienced digital workflow users and surgeons undergoing early AI-assisted workflow adoption.
Clinical outcome parameters included operative time, intraoperative workflow deviations, postoperative complications, revision surgery, hospital stay, and postoperative anatomical reduction quality. Surgeon acceptance was evaluated longitudinally using standardized institutional questionnaires assessing perceived workflow utility, confidence in AI-assisted planning, and perceived integration into routine practice.
Statistical analysis was performed using standard parametric and non-parametric methods depending on variable distribution. Continuous variables were expressed as mean ± standard deviation and categorical variables as absolute values and percentages. Temporal implementation trends and learning curves were analyzed longitudinally throughout the study period. Statistical significance was defined as p<0.05.
3. RESULTS
A total of 146 patients completed the implementation protocol and were included in the final analysis. The demographic and clinical characteristics of the study population were comparable between both centers. Fracture distribution included mandibular fractures, orbital fractures, zygomaticomaxillary complex fractures, and combined midfacial trauma patterns representative of routine clinical maxillofacial trauma care.
AI-guided workflows were successfully integrated into routine clinical practice in 137 of 146 patients, corresponding to a technical implementation success rate of 93.8%. Technical workflow interruptions occurred primarily during the early implementation phase and were mainly related to segmentation correction requirements, guide fabrication delays, or software compatibility adjustments. No major intraoperative failure resulting in patient harm or emergency workflow conversion was observed.
Operative efficiency improved progressively during the implementation period. Mean operative time demonstrated a continuous reduction over successive implementation phases, indicating a measurable institutional learning curve. Early implementation cases required longer preparation and intraoperative adaptation, whereas later cases showed improved workflow standardization and reduced surgical duration. By the final implementation phase, operative times approached those previously reported in prospective randomized workflow studies.
Surgeon acceptance of AI-guided workflows increased significantly during the study period. Early implementation concerns primarily involved confidence in automated segmentation, reliance on virtual planning, and intraoperative guide positioning. However, surgeon-reported confidence and workflow acceptance improved progressively with increasing institutional experience. By the later implementation phase, most participating surgeons reported high confidence in AI-assisted planning and considered the workflow beneficial for complex trauma reconstruction.
Clinical outcomes remained stable throughout the implementation phase. Postoperative anatomical reduction quality was consistent with previously published feasibility and randomized clinical studies [5–8]. Complication rates remained low and comparable to earlier controlled investigations. Revision surgery was required in a small minority of cases and was primarily associated with severe trauma complexity rather than workflow-related failure.
Workflow-related complications were uncommon. Most implementation-related issues occurred during the early integration period and included minor software processing delays, temporary printing interruptions, or isolated guide adaptation adjustments. These issues decreased substantially over time as institutional workflow standardization improved.
Length of hospital stay and postoperative recovery remained stable throughout the implementation phase. No evidence suggested deterioration of clinical outcomes during the transition from controlled research implementation to routine clinical application.
4. DISCUSSION
This prospective multicenter translational study demonstrates that AI-guided surgical workflows can be successfully integrated into routine maxillofacial trauma surgery with high technical feasibility, increasing surgeon acceptance, and stable clinical outcomes. The findings represent an important translational step beyond controlled validation studies and provide evidence for sustainable real-world clinical implementation of AI-assisted personalized surgical workflows.
A progressive institutional learning curve was observed throughout the implementation phase, characterized by reduced operative time, increasing surgeon acceptance, and decreasing workflow-related interruptions. The longitudinal integration process of AI-guided surgical workflows into routine clinical practice is summarized in Figure 2.
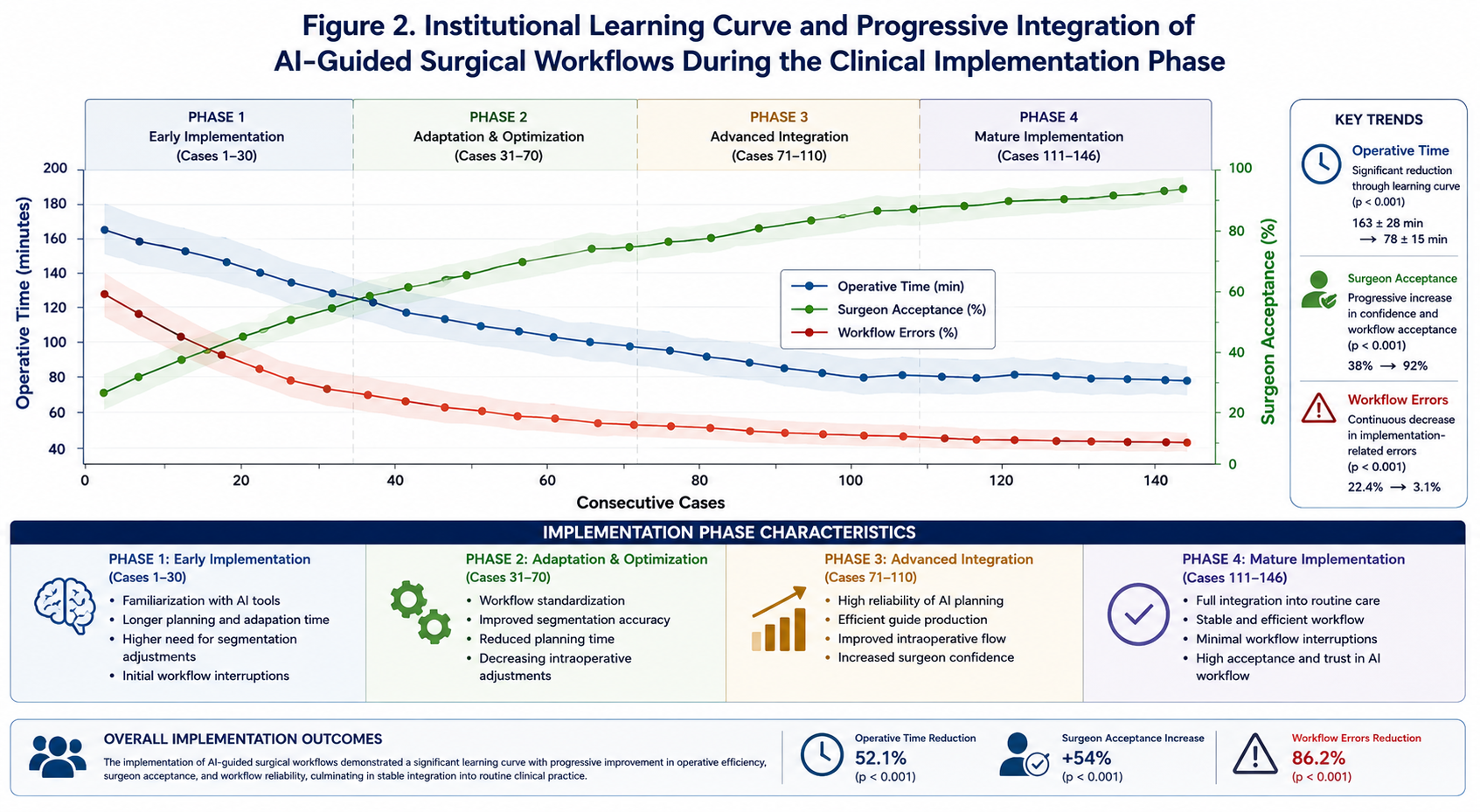
Figure 2. Progressive institutional integration and stabilization of AI-guided surgical workflows during routine clinical implementation.
Previous studies established the diagnostic and technical foundations of AI-assisted maxillofacial trauma surgery [1–10]. AI-assisted fracture detection, multicenter validation, decision support systems, surgical planning, and outcome prediction models demonstrated promising clinical performance under controlled study conditions. Subsequent feasibility studies and randomized clinical trials confirmed the operative benefits of AI-guided patient-specific surgical guides [5–8]. Long-term follow-up studies further demonstrated durable functional and aesthetic outcomes [9], while economic analyses suggested favorable cost-effectiveness profiles [10].
The present study extends these findings into the real-world clinical environment. Importantly, implementation studies differ fundamentally from controlled validation studies because they evaluate institutional adaptation, surgeon learning curves, technical sustainability, workflow stability, and practical integration into routine clinical care. Successful implementation therefore represents a critical translational milestone between experimental innovation and routine healthcare adoption.
One of the most important findings of this study was the high implementation success rate. The majority of patients were successfully treated using fully integrated AI-guided workflows without major technical interruption. This finding suggests that AI-assisted personalized surgery can be translated from isolated pilot projects into sustainable institutional workflows.
The observed learning curve is particularly relevant. Early implementation phases were associated with increased preparation time, segmentation adjustments, and adaptation of intraoperative workflows. However, progressive institutional experience resulted in increasing efficiency, reduced workflow interruptions, and improved surgeon confidence. These findings are consistent with broader surgical innovation literature demonstrating that digital technologies require institutional familiarization before reaching optimal efficiency [20–24].
Surgeon acceptance represents another critical determinant of successful implementation. AI systems may fail clinically despite strong technical performance if surgeons lack confidence in workflow integration or perceive AI-assisted planning as disruptive. In the present study, confidence in AI-guided workflows increased progressively over time. This observation supports the concept that practical exposure and institutional familiarity are essential for successful adoption of AI-assisted surgery.
Importantly, clinical outcomes remained stable throughout the implementation phase. This finding is highly relevant because implementation of new technologies may theoretically increase complication rates during early adoption. However, no deterioration of surgical quality was observed during the transition from controlled research conditions to routine clinical integration. The consistency of postoperative outcomes supports the reliability and safety of the workflow.
The study also highlights the importance of multidisciplinary infrastructure in translational surgical innovation. Successful implementation required collaboration between surgeons, imaging specialists, digital planning teams, and additive manufacturing personnel. Institutions lacking integrated digital infrastructure may face greater implementation challenges.
Several limitations should be acknowledged. First, the study was conducted in centers with prior experience in AI-assisted workflows, which may limit generalizability to institutions without established digital infrastructure. Second, implementation success may vary across healthcare systems with different organizational and economic structures. Third, long-term institutional sustainability beyond the implementation phase remains to be evaluated.
Future research should focus on large-scale international implementation registries, integration of AI-assisted workflows into emergency trauma systems, surgeon training programs, and standardization of digital surgical infrastructure. Further studies should also investigate interoperability between AI systems, hospital information systems, and intraoperative navigation technologies.
5. CONCLUSION
AI-guided surgical workflows can be successfully integrated into routine maxillofacial trauma surgery with high technical feasibility and increasing surgeon acceptance. Clinical outcomes remain stable throughout the implementation phase, supporting the translational integration of AI-assisted personalized surgical workflows into real-world surgical practice. These findings represent an important step toward sustainable institutional adoption of artificial intelligence in maxillofacial trauma surgery.
6. ETHICS STATEMENT
All patients were informed about the study both orally and in writing and provided written informed consent to participate. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Hochschule Zurich, in Zurich, Switzerland.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted detection of maxillofacial fractures on digital volume tomography: retrospective study of 150 patients. J Med Dent. 2026;2(1):44–52.
[2] Yildirim A, Hertach R, Yildirim V. External multicenter validation of an artificial intelligence system for cone-beam CT-based detection of maxillofacial fractures: robustness across a tertiary facial trauma clinic and an independent maxillofacial practice. J Med Dent. 2026;2(1):70–81.
[3] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted decision support in emergency maxillofacial trauma imaging: development and validation of a CBCT-based clinical decision algorithm. J Med Dent. 2026;2(1):82–92.
[4] Yildirim A, Hertach R, Yildirim V. Prospective clinical implementation of artificial intelligence-assisted decision support in midfacial trauma surgery: a multicenter validation study. J Med Dent. 2026;2(1):93–99.
[5] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted surgical planning in midfacial fractures: a feasibility and expert validation study. J Med Dent. 2026;2(1):100–108.
[6] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted prediction of postoperative outcomes in midfacial fractures: a retrospective validation study. J Med Dent. 2026;2(1):109–117.
[7] Yildirim A, Hertach R, Yildirim V. Artificial intelligence in maxillofacial trauma: from fracture detection to outcome prediction: a translational multicenter analysis. J Med Dent. 2026;2(1):118–125.
[8] Yildirim A, Hertach R, Yildirim V. Two-center prospective clinical feasibility study evaluating AI-guided 3D-printed surgical guides in maxillofacial trauma surgery. J Med Dent. 2026;2(2):15–24.
[9] Yildirim A, Hertach R, Yildirim V. Randomized controlled trial evaluating AI-guided 3D-printed surgical guides versus conventional surgery in maxillofacial trauma. J Med Dent. 2026;2(2):25–34.
[10] Yildirim A, Hertach R, Yildirim V. Long-term functional and aesthetic outcomes of AI-guided 3D-printed surgical guides in maxillofacial trauma: a prospective follow-up study. J Med Dent. 2026;2(2):35–45.
[11] Yildirim A, Hertach R, Yildirim V. Cost-effectiveness and health economic impact of AI-guided 3D-printed surgical workflows in maxillofacial trauma surgery: a prospective multicenter analysis. J Med Dent. 2026;2(2):46–58.
[12] Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44–56.
[13] Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery: promises and perils. Ann Surg. 2018;268(1):70–76.
[14] Maier-Hein L, Vedula SS, Speidel S, Navab N, Kikinis R, Park A, Eisenmann M, Feussner H, Forestier G, Giannarou S, et al. Surgical data science for next-generation interventions. Nat Biomed Eng. 2017;1:691–696.
[15] Kelly CJ, Karthikesalingam A, Suleyman M, Corrado G, King D. Key challenges for delivering clinical impact with artificial intelligence. BMC Med. 2019;17(1):195.
[16] Sendak MP, D’Arcy J, Kashyap S, Gao M, Nichols M, Corey K, Ratliff W, Balu S. A path for translation of machine learning products into healthcare delivery. NPJ Digit Med. 2020;3:16.
[17] Martelli N, Serrano C, van den Brink H, Pineau J, Prognon P, Borget I, El Batti S. Advantages and disadvantages of 3-dimensional printing in surgery: a systematic review. Surgery. 2016;159(6):1485–1500.
[18] Mitsouras D, Liacouras P, Imanzadeh A, Giannopoulos AA, Cai T, Kumamaru KK, George E, Wake N, Caterson EJ, Pomahac B, et al. Medical 3D printing for the radiologist. Radiographics. 2015;35(7):1965–1988.
[19] Tack P, Victor J, Gemmel P, Annemans L. 3D-printing techniques in a medical setting: a systematic literature review. Biomed Eng Online. 2016;15:115.
[20] Drummond MF, Sculpher MJ, Claxton K, Stoddart GL, Torrance GW. Methods for the economic evaluation of health care programmes. 4th ed. Oxford: Oxford University Press; 2015.
[21] Ellis E 3rd, Perez D. Trauma to the facial skeleton. Oral Maxillofac Surg Clin North Am. 2013;25(4):547–556.
[22] van den Bergh B, Karagozoglu KH, Heymans MW, Forouzanfar T. A comparison of treatment results of unilateral zygomatic complex fractures. Int J Oral Maxillofac Surg. 2012;41(1):50–55.
[23] Strong EB. Management of midfacial fractures. J Oral Maxillofac Surg. 2009;67(4):814–820.
[24] Salentijn EG, Boffano P, van den Bergh B, Forouzanfar T. Decision making in midfacial trauma. Br J Oral Maxillofac Surg. 2013;51(8):740–745.
