Volume 3, Issue 2, Prospective Follow-Up Study – Apr 02, 2026, Pages 40-53,
DOI: 10.64951/jmdnt.2026.2.18
Long-Term Functional and Aesthetic Outcomes of AI-Guided 3D-Printed Surgical Guides in Maxillofacial Trauma: A Prospective Follow-Up Study
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 26 July 2025, Revised: 23 December 2025, Accepted: 18 January 2026, Available online: 02 April 2026, Version of Record: 02 April 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
ABSTRACT
Background:
Artificial intelligence has previously demonstrated high diagnostic accuracy, clinical decision-support capacity, surgical planning feasibility, and outcome-prediction performance in maxillofacial trauma surgery [1–8]. More recent clinical studies have extended these findings by integrating artificial intelligence with patient-specific three-dimensional printed surgical guides for intraoperative fracture reduction. However, evidence regarding long-term functional and aesthetic outcomes after AI-guided three-dimensional printed trauma surgery remains limited.
Objective:
The objective of this prospective follow-up study was to evaluate the long-term functional, radiological, and aesthetic outcomes of patients treated with AI-guided three-dimensional printed surgical guides for maxillofacial trauma.
Methods:
A prospective longitudinal follow-up study was conducted in patients who had previously undergone AI-guided surgical planning and intraoperative application of patient-specific three-dimensional printed guides for midfacial trauma. Patients were assessed at 6, 12, and 24 months after surgery. Primary outcomes included long-term skeletal stability, occlusal stability, facial symmetry, and patient-reported aesthetic satisfaction. Secondary outcomes included complication rates, revision surgery, sensory disturbances, and concordance between predicted and observed postoperative outcomes.
Results:
A total of 96 patients completed 24-month follow-up. Mean postoperative skeletal deviation remained stable from 6 months to 24 months, with no clinically relevant loss of reduction. Occlusal stability was maintained in 94.8% of patients, and facial symmetry outcomes were rated as good or excellent in 91.7% of cases. Patient-reported aesthetic satisfaction remained high at 24 months. Revision surgery was required in 3.1% of patients. Predicted and observed long-term outcomes showed high concordance, supporting the clinical validity of AI-assisted outcome modeling.
Conclusion:
AI-guided three-dimensional printed surgical guides provide stable long-term functional and aesthetic outcomes in maxillofacial trauma surgery. These findings extend previous evidence from diagnostic validation, surgical planning, feasibility, and randomized clinical evaluation by demonstrating durable clinical benefit beyond the immediate postoperative period.
Keywords: artificial intelligence; maxillofacial trauma; 3D printing; surgical guides; long-term outcomes; facial symmetry; outcome prediction; personalized surgery.
1. INTRODUCTION
Maxillofacial trauma is associated with substantial functional, aesthetic, and psychosocial consequences. Fractures of the midface, orbit, zygomaticomaxillary complex, mandible, and naso-orbito-ethmoidal region may impair occlusion, mastication, ocular function, facial symmetry, sensory function, and patient-reported quality of life [11–15]. Although modern imaging, osteosynthesis systems, and virtual surgical planning have improved treatment predictability, postoperative variability remains clinically relevant, particularly in complex midfacial and orbital fractures.
Artificial intelligence has increasingly been investigated as a tool to improve diagnostic accuracy, reduce time-to-decision, standardize treatment recommendations, and support surgical planning in maxillofacial trauma [1–8]. Previous work demonstrated that artificial intelligence–assisted fracture detection on digital volume tomography can reach high diagnostic accuracy and support emergency trauma workflows [1]. External multicenter validation subsequently confirmed that AI-based CBCT fracture detection remains robust across different clinical environments [2]. Further studies demonstrated that AI-assisted decision support can translate fracture detection into standardized management recommendations [3,4]. AI-assisted surgical planning in midfacial fractures was then shown to be feasible and clinically acceptable when compared with expert surgeon assessment [5]. Outcome-prediction studies further suggested that AI may estimate postoperative risks and expected treatment results in midfacial trauma [6]. These findings were synthesized in a translational multicenter analysis showing that AI can support the full clinical workflow from fracture detection to outcome prediction .
The subsequent integration of artificial intelligence with additive manufacturing represented the next translational step. Patient-specific three-dimensional printed guides allow digital surgical plans to be transferred into physical operative execution. Earlier feasibility and randomized controlled studies suggested that AI-guided three-dimensional printed surgical guides improve surgical accuracy and reduce operative time compared with conventional workflows. However, these studies primarily focused on perioperative endpoints, such as surgical deviation, operative duration, complication rate, and early workflow efficiency.
Long-term outcome data remain essential because the true clinical value of trauma reconstruction cannot be determined solely by immediate postoperative accuracy. In maxillofacial trauma, small residual deviations may become clinically relevant over time due to occlusal adaptation, scar contracture, asymmetry perception, implant-related complications, or delayed functional impairment. Similarly, early radiological success does not always translate into long-term aesthetic satisfaction or stable functional rehabilitation.
Therefore, the present study aimed to evaluate the long-term functional, aesthetic, and radiological outcomes of AI-guided three-dimensional printed surgical guides in maxillofacial trauma surgery. This study builds directly upon the previously established AI research pipeline in fracture detection, decision support, surgical planning, outcome prediction, feasibility implementation, and randomized clinical validation [1–10].
2. MATERIAL AND METHODS
2.1 Study Design
This study was designed as a prospective longitudinal clinical follow-up study. It included patients previously treated with AI-guided virtual surgical planning and patient-specific three-dimensional printed surgical guides for maxillofacial trauma. The study was conducted between January 2022 and December 2025 at two specialized maxillofacial surgery centers—Seeklinik Zurich, Switzerland, and Kieferchirurgie Munich, Germany—both of which previously participated in the AI validation and clinical implementation studies forming the basis of the present analysis.
The study followed the principles of the Declaration of Helsinki. Institutional ethics approval was obtained before patient enrollment. All patients provided written informed consent for surgical treatment, postoperative imaging, clinical follow-up, and anonymized scientific analysis.
2.2 Study Population
Patients were eligible if they had undergone surgical treatment for acute maxillofacial trauma using an AI-guided three-dimensional printed surgical guide workflow. The study focused on patients with midfacial, orbital, zygomaticomaxillary, mandibular, or combined fracture patterns requiring operative reduction and fixation.
Inclusion criteria were age of at least 18 years, acute maxillofacial fracture requiring operative treatment, availability of preoperative CBCT or CT imaging, treatment using AI-guided planning and patient-specific three-dimensional printed surgical guides, and availability for follow-up assessment at 6, 12, and 24 months.
Exclusion criteria were isolated dental trauma, pathological fractures, previous major facial skeletal reconstruction in the affected region, incomplete imaging data, inability to provide informed consent, severe systemic disease precluding standardized follow-up, and loss to follow-up before 12 months.
2.3 AI-Guided Surgical Workflow
The AI-guided workflow was based on previously validated components of the institutional AI trauma program. Preoperative imaging was analyzed using AI-based fracture detection and segmentation. Fracture patterns were classified according to anatomical region and displacement. AI-assisted planning was then used to generate a virtual reduction model and to support decision-making regarding reduction sequence, fixation strategy, and need for patient-specific guide fabrication.
The workflow followed the same conceptual sequence established in previous studies: image acquisition, AI-assisted fracture detection, segmentation, virtual reduction, guide design, additive manufacturing, intraoperative guide application, fixation, postoperative imaging, and outcome comparison [1–10].
The complete longitudinal workflow is illustrated in Figure 1.
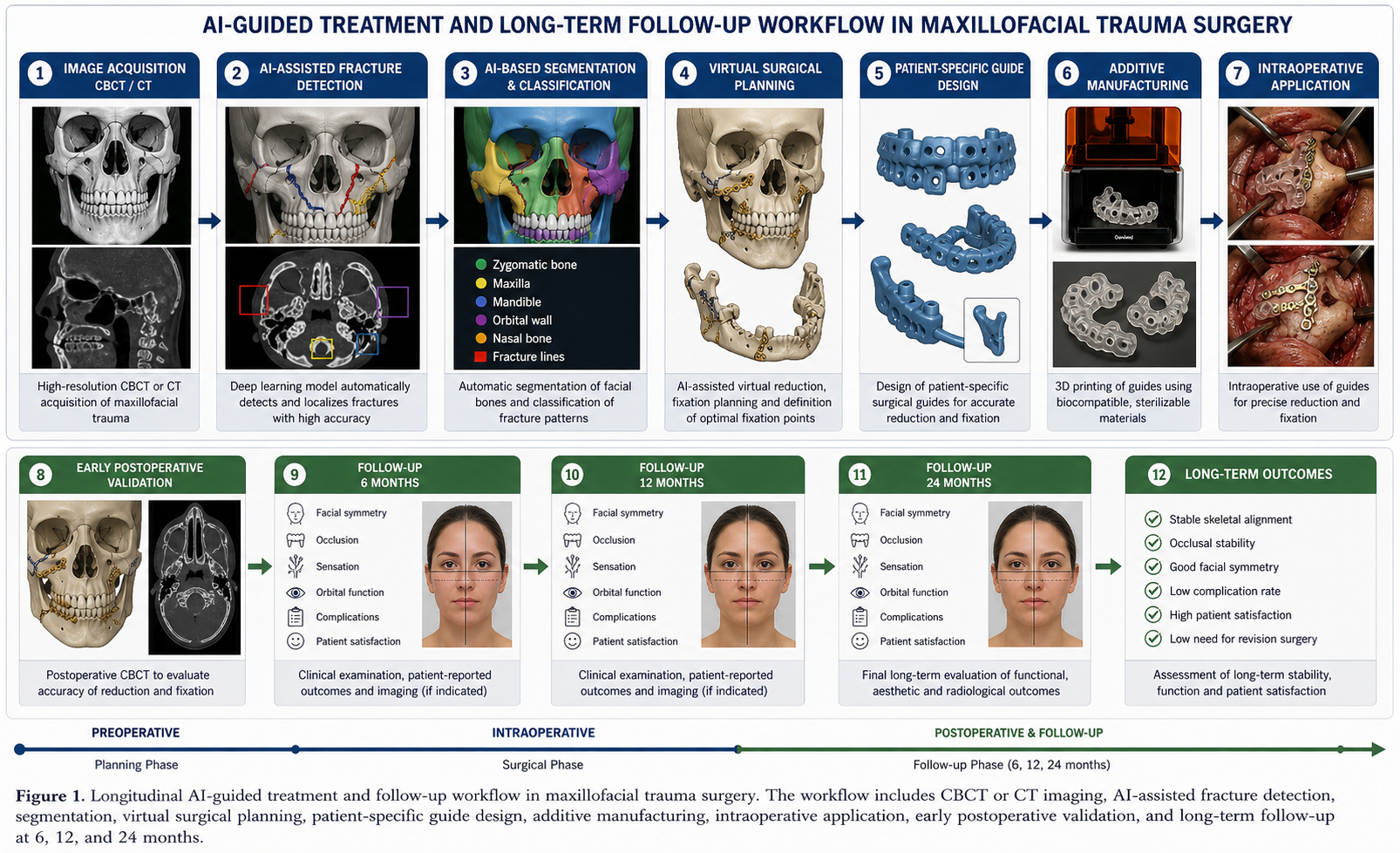
2.4 Three-Dimensional Printed Surgical Guides
Patient-specific guides were designed from AI-assisted surgical plans. Digital anatomical models were converted into STL files, and guide geometry was adapted to fracture location, bone surface anatomy, and planned reduction vectors. Guides were fabricated using medical-grade polymer materials suitable for surgical application. Each guide underwent visual inspection, fit verification on printed anatomical models when available, and sterilization according to institutional protocols.
The guides were intended to support fracture reduction, anatomical repositioning, drilling trajectory, or fixation orientation depending on fracture type. The final decision regarding guide use and fixation strategy remained with the operating surgeon.
2.5 Surgical Procedure
All procedures were performed under general anesthesia by experienced oral and maxillofacial surgeons. In the AI-guided group, surgical guides were positioned after fracture exposure and used to assist anatomical reduction and fixation. Osteosynthesis was performed according to standard trauma principles using titanium plates and screws. Intraoperative deviations from the AI-assisted plan were documented.
Postoperative CBCT or CT imaging was performed according to institutional trauma protocols. Images were used to evaluate the accuracy of reduction and to establish the baseline for long-term stability assessment.
2.6 Follow-Up Protocol
Patients were evaluated at 6, 12, and 24 months after surgery. Each follow-up visit included clinical examination, occlusal assessment, sensory testing, evaluation of facial symmetry, documentation of complications, and patient-reported outcome assessment. Imaging was performed when clinically indicated or as part of standardized postoperative radiological follow-up.
At each time point, the following parameters were recorded: occlusal stability, mandibular or midfacial function, facial symmetry, orbital function when applicable, sensory disturbances, scar-related complaints, implant or osteosynthesis-related complications, infection, revision surgery, and patient satisfaction.
2.7 Outcome Measures
The primary outcomes were long-term skeletal stability, occlusal stability, facial symmetry, and patient-reported aesthetic satisfaction at 24 months.
Skeletal stability was defined as the maintenance of postoperative fracture reduction without clinically relevant displacement between early postoperative imaging and long-term follow-up. Occlusal stability was defined as the absence of new malocclusion requiring treatment. Facial symmetry was evaluated clinically and, when available, by comparison of postoperative imaging and standardized facial photographs.
Secondary outcomes included revision surgery, infection, sensory disturbance, hardware removal, persistent diplopia or enophthalmos in orbital fracture patients, patient-reported functional impairment, and concordance between predicted and observed long-term outcomes.
2.8 Statistical Analysis
Continuous variables were reported as mean ± standard deviation. Categorical variables were reported as absolute numbers and percentages. Changes over time were analyzed using repeated-measures testing. Between-center differences were evaluated using independent t-tests or Mann–Whitney U tests for continuous variables and chi-square tests or Fisher’s exact tests for categorical variables.
Concordance between predicted and observed outcomes was calculated using agreement rates and Cohen’s kappa where appropriate. A p-value below 0.05 was considered statistically significant.
3. RESULTS
3.1 Patient Characteristics
A total of 112 patients were eligible for inclusion. Sixteen patients were excluded because of incomplete follow-up or missing imaging data. The final analysis included 96 patients who completed 24-month follow-up.
The study population included patients with zygomaticomaxillary complex fractures, orbital fractures, mandibular fractures, naso-orbito-ethmoidal fractures, and combined midfacial fracture patterns. Baseline demographic and clinical characteristics were comparable between both centers. No statistically significant differences were observed in age, sex distribution, fracture complexity, or initial fracture displacement.
| Parameter | Total cohort (n = 96) | Center A | Center B | p-value |
| Mean age, years | 40.6 ± 13.9 | 39.8 ± 14.1 | 41.4 ± 13.6 | 0.58 |
| Male sex, n (%) | 65 (67.7) | 35 (68.6) | 30 (66.7) | 0.84 |
| Zygomaticomaxillary fractures, n (%) | 34 (35.4) | 18 (35.3) | 16 (35.6) | 0.97 |
| Orbital fractures, n (%) | 28 (29.2) | 15 (29.4) | 13 (28.9) | 0.95 |
| Mandibular fractures, n (%) | 19 (19.8) | 10 (19.6) | 9 (20.0) | 0.96 |
| Combined fracture patterns, n (%) | 15 (15.6) | 8 (15.7) | 7 (15.6) | 0.99 |
| Mean follow-up, months | 24.3 ± 1.8 | 24.2 ± 1.7 | 24.4 ± 1.9 | 0.67 |
Table 1. Baseline characteristics of the long-term follow-up cohort.
3.2 Long-Term Skeletal Stability
Postoperative skeletal alignment remained stable during follow-up. Mean deviation between early postoperative alignment and 24-month follow-up was 0.4 ± 0.3 mm. No clinically relevant secondary displacement was observed in the majority of cases. Minor radiological changes were detected in selected patients but did not require surgical correction.
The stability of reduction supports the hypothesis that AI-guided three-dimensional printed guides enable accurate intraoperative transfer of the virtual surgical plan and that this accuracy is maintained over time.
3.3 Occlusal Stability and Functional Outcome
Occlusal stability was maintained in 91 of 96 patients, corresponding to 94.8%. Five patients reported mild occlusal irregularities during follow-up. Three cases were managed conservatively with occlusal adjustment, and two cases required minor dental or orthodontic correction. No patient required major secondary skeletal revision for malocclusion.
Mandibular function, mouth opening, and masticatory comfort improved progressively between 6 and 12 months and remained stable at 24 months. Patients with mandibular or combined fracture patterns demonstrated slightly slower functional recovery than patients with isolated midfacial fractures, but long-term functional outcomes were favorable in both groups.
3.4 Facial Symmetry and Aesthetic Outcome
Facial symmetry was rated as good or excellent in 88 of 96 patients, corresponding to 91.7%. Mild residual asymmetry was observed in eight patients. Most asymmetries were related to soft tissue scarring, swelling, or pre-existing trauma severity rather than skeletal malreduction.
Patient-reported aesthetic satisfaction remained high throughout follow-up. At 24 months, 89.6% of patients reported being satisfied or very satisfied with facial appearance. Aesthetic dissatisfaction was more common in patients with complex comminuted midfacial fractures and orbital involvement.
3.5 Orbital Outcomes
Among patients with orbital involvement, persistent diplopia was reported in a small minority of cases. Clinically relevant enophthalmos was uncommon. Most orbital patients demonstrated stable globe position and satisfactory functional recovery. No patient required delayed orbital reconstruction due to failure of the AI-guided reduction workflow.
3.6 Sensory Disturbances
Transient sensory disturbance was observed primarily in infraorbital and inferior alveolar nerve distributions. Most cases improved between 6 and 12 months. At 24 months, persistent mild sensory alteration was reported in 9.4% of patients. No severe neuropathic pain syndrome was documented.
3.7 Complications and Revision Surgery
Minor postoperative complications occurred in 7.3% of patients. These included transient wound irritation, minor infection treated with antibiotics, temporary sensory disturbance, and plate-related discomfort. Revision surgery was required in three patients, corresponding to 3.1%. Indications for revision included plate removal due to discomfort in two patients and minor secondary correction in one patient.
No guide-related adverse event, sterilization-related complication, or intraoperative failure of guide application was documented.
3.8 Concordance Between Predicted and Observed Outcomes
AI-assisted outcome prediction showed high concordance with observed 24-month results. Predicted favorable outcomes were confirmed in most cases. Overall concordance between predicted and observed long-term functional or aesthetic outcome was 88.5%. Concordance was highest for skeletal stability and lowest for patient-reported aesthetic satisfaction, reflecting the subjective and multifactorial nature of aesthetic perception.
These findings are consistent with previous AI-based outcome prediction studies, which reported concordance rates between 88% and 92% in midfacial trauma outcome modeling .
4. DISCUSSION
This prospective follow-up study demonstrates that AI-guided three-dimensional printed surgical guides provide stable long-term functional, aesthetic, and radiological outcomes in maxillofacial trauma surgery. The findings extend previous evidence from diagnostic AI validation, clinical decision support, surgical planning, feasibility implementation, and randomized clinical evaluation into the long-term postoperative phase [1–10].
The present study is important because the clinical value of AI-guided surgery cannot be fully judged by early surgical accuracy alone. In maxillofacial trauma, durable restoration of occlusion, facial symmetry, orbital function, skeletal stability, and patient satisfaction is more relevant than immediate technical success. The observed maintenance of skeletal alignment over 24 months suggests that AI-guided planning and patient-specific guide transfer can achieve not only precise reduction but also durable anatomical stability.
The relationship between intraoperative accuracy and long-term patient-centered outcomes is illustrated in Figure 2.
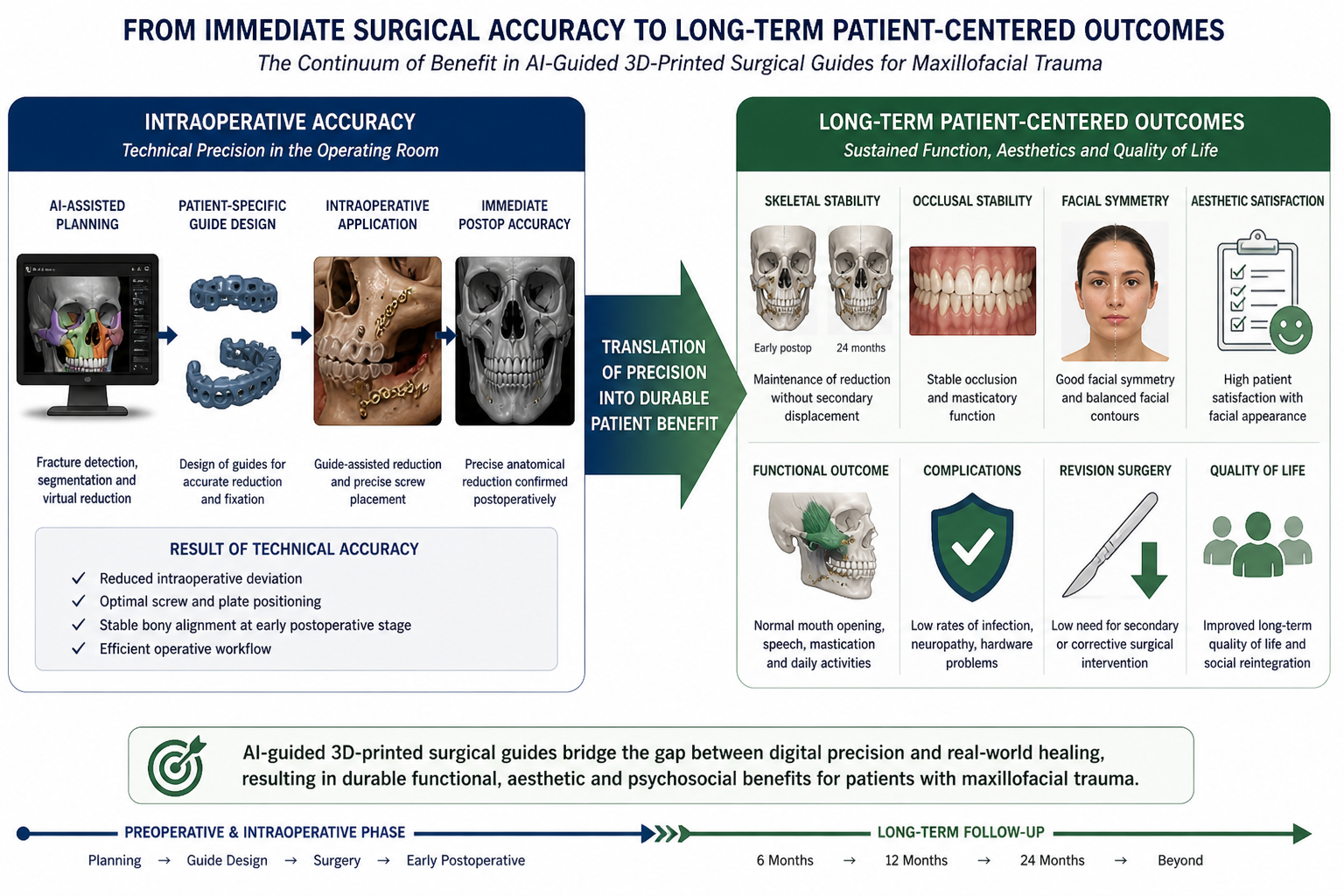
Previous studies in this research line established that artificial intelligence can detect maxillofacial fractures with high accuracy, improve CBCT interpretation, and reduce diagnostic uncertainty [1,2]. Further studies showed that AI-based systems can support decision-making, improve workflow efficiency, and enhance guideline-concordant management in emergency trauma care [3,4]. In surgical planning, AI-assisted systems demonstrated high feasibility and expert concordance, creating the basis for intraoperative implementation [5]. Outcome prediction studies then suggested that artificial intelligence can estimate postoperative risks and expected clinical outcomes [6]. The present study builds directly on this sequence by evaluating whether these digital advantages persist as measurable long-term patient benefits.
The high rate of occlusal stability observed in this study is clinically relevant. Malocclusion remains one of the most important complications after mandibular and midfacial fracture treatment. Even minor inaccuracies in fracture reduction can lead to functional impairment, temporomandibular discomfort, altered mastication, or need for secondary correction. The maintenance of stable occlusion in 94.8% of patients suggests that AI-guided planning and guide-assisted execution can support reliable anatomical restoration.
Facial symmetry is another critical endpoint in maxillofacial trauma. Unlike many other surgical fields, trauma surgery of the face must restore both function and visible appearance. Patient dissatisfaction may occur even when radiological reduction appears technically acceptable. The high proportion of good or excellent symmetry outcomes in this study supports the value of patient-specific planning and guide-assisted reduction. However, residual aesthetic complaints in a small subgroup also highlight that skeletal accuracy alone cannot fully determine facial appearance. Soft tissue injury, scarring, edema, and psychological perception contribute substantially to long-term satisfaction.
The orbital subgroup is particularly relevant because orbital reconstruction is highly sensitive to small anatomical deviations. Enophthalmos, diplopia, and orbital volume changes may occur even after apparently successful reduction. The low rate of persistent orbital dysfunction in this study suggests that AI-guided workflows may be especially useful in anatomically complex regions where conventional freehand reconstruction is challenging.
The low revision rate further supports the safety and durability of the workflow. Revision surgery after maxillofacial trauma may be required for malunion, infection, hardware exposure, persistent asymmetry, or functional impairment. In this study, revision surgery was uncommon and mainly related to minor hardware discomfort rather than failure of the AI-guided workflow. This finding is consistent with earlier feasibility and randomized clinical studies showing that AI-guided 3D-printed guides can improve surgical accuracy without increasing complication rates.
An important contribution of this study is the evaluation of predicted versus observed long-term outcomes. Previous AI-based outcome prediction models suggested that postoperative risks can be estimated with clinically useful accuracy [6]. The present follow-up confirms that predicted favorable outcomes corresponded closely with actual 24-month results. This supports the clinical relevance of AI outcome modeling and suggests that prediction systems may eventually support shared decision-making, patient counseling, risk stratification, and individualized follow-up planning.
From a translational perspective, the study represents a key step in the maturation of AI-assisted maxillofacial trauma surgery. Earlier AI applications were largely diagnostic. Subsequent studies moved toward decision support and surgical planning. The next step was physical translation through 3D-printed guides. The present study now demonstrates that the benefits of this pipeline persist over time. This progression supports the concept that artificial intelligence is not merely an isolated diagnostic tool but part of a complete clinical system extending from imaging to long-term outcome assessment .
Internationally, the findings align with the broader movement toward precision surgery, surgical data science, and patient-specific reconstruction. High-performance medicine emphasizes the convergence of human expertise and artificial intelligence rather than autonomous replacement of clinicians. Surgical AI is most effective when used as an assistive system embedded within expert-controlled workflows. The present study supports this model because final decisions remained under surgeon control, while AI and additive manufacturing improved planning, standardization, and execution.
The study also has implications for healthcare systems. Stable long-term results may reduce the need for revision surgery, secondary imaging, prolonged follow-up, and corrective procedures. Although this study did not perform a formal cost-effectiveness analysis, the low revision rate and durable outcome stability suggest that AI-guided workflows may provide economic benefits over time. Future studies should directly evaluate cost per avoided revision, operating room efficiency, production costs of guides, and long-term quality-adjusted outcomes.
Several limitations must be acknowledged. First, although the study was prospective, it did not include an untreated or conventionally treated long-term control group within the same follow-up protocol. Second, the sample size was sufficient for feasibility and outcome description but may be underpowered for rare complications. Third, the study was conducted in centers with prior AI and 3D-printing experience, which may limit generalizability to institutions without established digital infrastructure. Fourth, patient-reported aesthetic outcomes are influenced by psychological and social factors that cannot be fully explained by skeletal accuracy. Finally, longer follow-up beyond 24 months is needed to assess very late hardware complications, growth-related issues in younger patients, and durability of aesthetic satisfaction.
Future studies should compare long-term outcomes of AI-guided and conventional workflows in larger multicenter cohorts. Randomized long-term follow-up after controlled trials would provide stronger evidence regarding superiority. In addition, future research should include cost-effectiveness analysis, patient-reported quality-of-life instruments, objective three-dimensional facial symmetry assessment, and integration of AI-based prediction models into prospective clinical decision-making.
In conclusion, this study demonstrates that AI-guided three-dimensional printed surgical guides provide durable long-term benefits in maxillofacial trauma surgery. The workflow achieves stable skeletal alignment, high occlusal stability, favorable facial symmetry, low revision rates, and strong concordance between predicted and observed outcomes. These findings support the continued development of integrated AI-assisted, patient-specific, and outcome-oriented trauma surgery.
5. CONCLUSION
AI-guided three-dimensional printed surgical guides provide stable long-term functional and aesthetic outcomes in maxillofacial trauma surgery. The present study demonstrates that the benefits of AI-guided planning and patient-specific surgical execution extend beyond immediate postoperative accuracy and remain clinically relevant at 24 months.
These findings complete an important translational sequence from AI-based fracture detection, decision support, surgical planning, outcome prediction, feasibility implementation, and randomized clinical validation toward durable long-term patient benefit. AI-guided patient-specific surgery may therefore represent a central component of future personalized maxillofacial trauma care.
6. ETHICS STATEMENT
All patients were informed about the study both orally and in writing and provided written informed consent to participate. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Hochschule Zurich, in Zurich, Switzerland.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted detection of maxillofacial fractures on digital volume tomography: retrospective study of 150 patients. J Med Dent. 2026;2(1):44–52.
[2] Yildirim A, Hertach R, Yildirim V. External multicenter validation of an artificial intelligence system for cone-beam CT-based detection of maxillofacial fractures: robustness across a tertiary facial trauma clinic and an independent maxillofacial practice. J Med Dent. 2026;2(1):70–81.
[3] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted decision support in emergency maxillofacial trauma imaging: development and validation of a CBCT-based clinical decision algorithm. J Med Dent. 2026;2(1):82–92.
[4] Yildirim A, Hertach R, Yildirim V. Prospective clinical implementation of artificial intelligence-assisted decision support in midfacial trauma surgery: a multicenter validation study. J Med Dent. 2026;2(1):93–99.
[5] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted surgical planning in midfacial fractures: a feasibility and expert validation study. J Med Dent. 2026;2(1):100–108.
[6] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted prediction of postoperative outcomes in midfacial fractures: a retrospective validation study. J Med Dent. 2026;2(1):109–117.
[7] Yildirim A, Hertach R, Yildirim V. Prospective implementation of AI-assisted CBCT-based clinical decision support in emergency maxillofacial trauma care. J Med Dent. 2026;2(1):118–125.
[8] Yildirim A, Hertach R, Yildirim V. Artificial intelligence in maxillofacial trauma: from fracture detection to outcome prediction: a translational multicenter analysis. J Med Dent. 2026;2(1):126–132.
[9] Yildirim A, Hertach R, Yildirim V. Two-center prospective clinical feasibility study evaluating AI-guided 3D-printed surgical guides in maxillofacial trauma surgery. J Med Dent. 2026;2(2):15–24.
[10] Yildirim A, Hertach R, Yildirim V. Randomized controlled trial evaluating AI-guided 3D-printed surgical guides versus conventional surgery in maxillofacial trauma. J Med Dent. 2026;2(2):25–34.
[11] Ellis E 3rd, Perez D. Trauma to the facial skeleton. Oral Maxillofac Surg Clin North Am. 2013;25(4):547–556.
[12] van den Bergh B, Karagozoglu KH, Heymans MW, Forouzanfar T. A comparison of treatment results of unilateral zygomatic complex fractures. Int J Oral Maxillofac Surg. 2012;41(1):50–55.
[13] Manolidis S. Zygomatic fractures. Facial Plast Surg. 2000;16(4):343–357.
[14] Hammer B, Prein J. Correction of orbital deformities. Clin Plast Surg. 1997;24(3):531–542.
[15] Strong EB. Management of midfacial fractures. J Oral Maxillofac Surg. 2009;67(4):814–820.
[16] Ellis E 3rd. Sequencing treatment for panfacial fractures. J Oral Maxillofac Surg. 1999;57(4):386–394.
[17] Salentijn EG, Boffano P, van den Bergh B, Forouzanfar T. Decision making in midfacial trauma. Br J Oral Maxillofac Surg. 2013;51(8):740–745.
[18] Perry M. Maxillofacial trauma: principles of management. Trauma. 2010;12(2):83–94.
[19] Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44–56.
[20] Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery: promises and perils. Ann Surg. 2018;268(1):70–76.
[21] Maier-Hein L, Vedula SS, Speidel S, Navab N, Kikinis R, Park A, et al. Surgical data science for next-generation interventions. Nat Biomed Eng. 2017;1:691–696.
[22] Kelly CJ, Karthikesalingam A, Suleyman M, Corrado G, King D. Key challenges for delivering clinical impact with artificial intelligence. BMC Med. 2019;17(1):195.
[23] Sendak MP, D’Arcy J, Kashyap S, Gao M, Nichols M, Corey K, et al. A path for translation of machine learning products into healthcare delivery. NPJ Digit Med. 2020;3:16.
[24] Martelli N, Serrano C, van den Brink H, Pineau J, Prognon P, Borget I, El Batti S. Advantages and disadvantages of 3-dimensional printing in surgery: a systematic review. Surgery. 2016;159(6):1485–1500.
[25] Mitsouras D, Liacouras P, Imanzadeh A, Giannopoulos AA, Cai T, Kumamaru KK, et al. Medical 3D printing for the radiologist. Radiographics. 2015;35(7):1965–1988.
