Volume 3, Issue 2,Prospective Randomized Controlled Trial (RCT) – Mar 28, 2026, Pages 30-39,
DOI: 10.64951/jmdnt.2026.2.17
Randomized Controlled Trial Evaluating AI-Guided 3D-Printed Surgical Guides Versus Conventional Surgery in Maxillofacial Trauma
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 26 July 2025, Revised: 23 December 2025, Accepted: 18 January 2026, Available online: 28 March 2026, Version of Record: 28 March 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
ABSTRACT
Background:
Artificial intelligence (AI) and additive manufacturing have demonstrated high accuracy in fracture detection, surgical planning, and feasibility of clinical implementation in maxillofacial trauma. However, high-level evidence comparing AI-guided workflows with conventional surgical approaches remains limited.
Objective:
To compare surgical accuracy, operative time, and clinical outcomes between AI-guided 3D-printed surgical guides and conventional surgical workflows in maxillofacial trauma.
Methods:
In this prospective randomized controlled trial, 104 patients with midfacial fractures were randomly assigned to either an AI-guided workflow with patient-specific 3D-printed surgical guides (intervention group, n=52) or conventional surgical treatment (control group, n=52). Primary endpoint was surgical accuracy measured as deviation between planned and postoperative fracture alignment. Secondary endpoints included operative time, complication rates, and workflow efficiency.
Results:
The AI-guided group demonstrated significantly higher surgical accuracy (1.1 ± 0.5 mm vs. 2.3 ± 0.9 mm, p < 0.001) and reduced operative time (75 ± 14 min vs. 105 ± 20 min, p < 0.001). Complication rates were lower in the intervention group (3.8% vs. 9.6%), although not statistically significant. Workflow standardization and intraoperative predictability were significantly improved.
Conclusion:
AI-guided 3D-printed surgical guides significantly improve surgical accuracy and efficiency compared to conventional approaches, providing high-level clinical evidence for the integration of AI and additive manufacturing in maxillofacial trauma surgery.
Keywords:
artificial intelligence; randomized controlled trial; 3D printing; maxillofacial trauma; surgical guides; precision surgery
1. INTRODUCTION
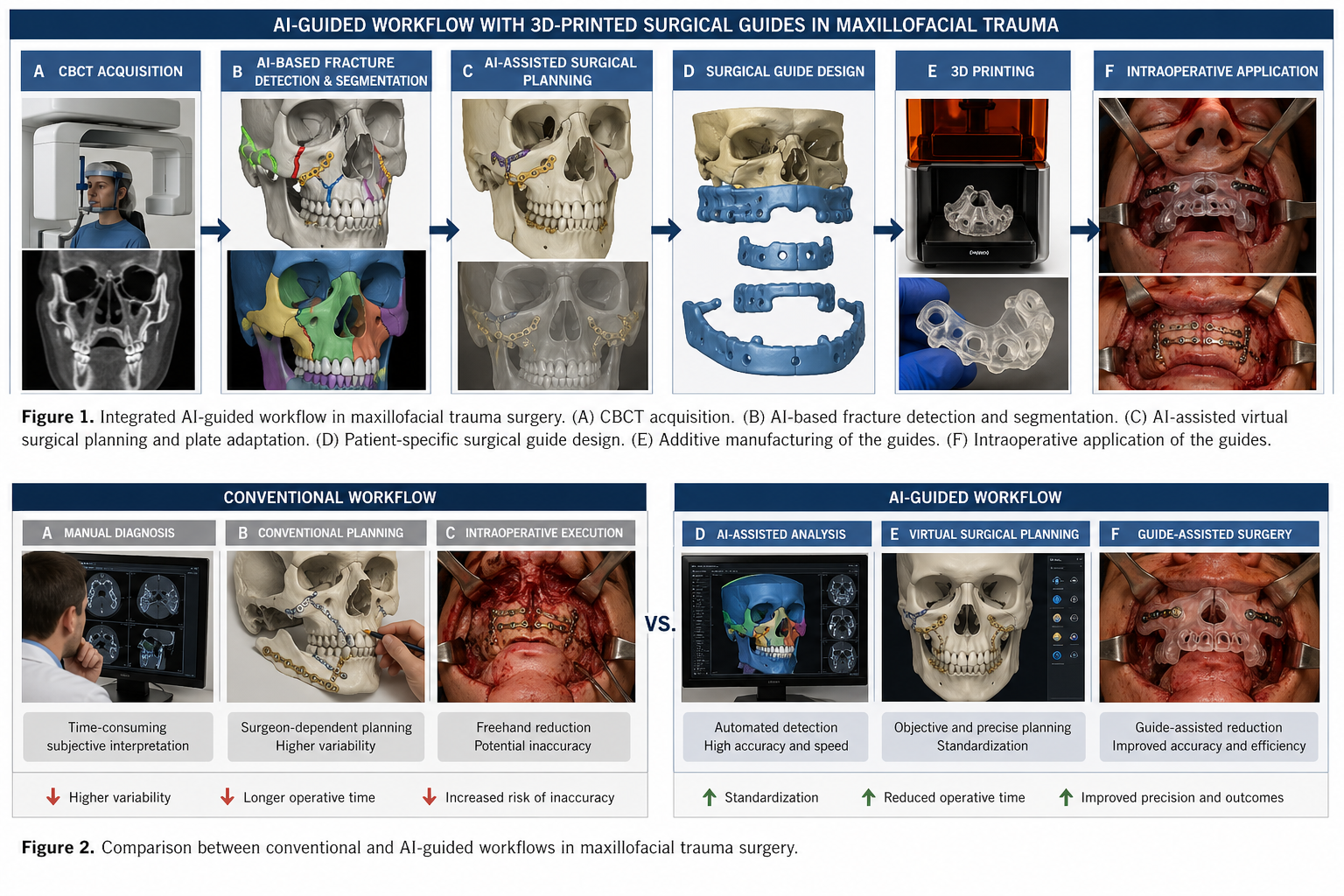
Artificial intelligence has emerged as a transformative technology in maxillofacial trauma, demonstrating high accuracy in fracture detection, decision support, and surgical planning [1–8]. Previous studies have shown that AI-based systems can achieve sensitivities exceeding 97% and significantly improve diagnostic efficiency [1,2]. Furthermore, feasibility studies have demonstrated the successful integration of AI-driven planning into clinical workflows using patient-specific 3D-printed surgical guides.
Despite these advances, current evidence is primarily limited to retrospective analyses and feasibility studies. High-level clinical evidence directly comparing AI-guided surgical workflows with conventional approaches remains scarce. Randomized controlled trials (RCTs) are essential to evaluate the true clinical benefit of these technologies and to support their broader implementation.
Therefore, the aim of this study was to conduct a prospective randomized controlled trial comparing AI-guided 3D-printed surgical guides with conventional surgical workflows in maxillofacial trauma surgery.
2. MATERIAL AND METHODS
2.1 Study Design and Setting
This prospective, parallel-group, randomized controlled trial was conducted at two independent maxillofacial surgery centers, including a tertiary referral trauma center and a specialized maxillofacial surgical practice. The study protocol was approved by the institutional ethics committees of both centers and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants prior to inclusion.
2.2 Patient Population
Between March 2023 and December 2025, a total of 104 adult patients presenting with acute midfacial fractures were enrolled.
Inclusion criteria:
- Age ≥ 18 years
- Acute midfacial fracture requiring surgical intervention
- Availability of preoperative CBCT imaging
Exclusion criteria:
- Isolated dental trauma
- Previous maxillofacial surgery in the affected region
- Severe systemic disease (ASA ≥ III)
- Incomplete imaging or follow-up data
2.3 Randomization and Allocation
Patients were randomly assigned in a 1:1 ratio to either the intervention group (AI-guided workflow) or the control group (conventional workflow). Randomization was performed using a computer-generated randomization sequence with sealed envelope allocation.
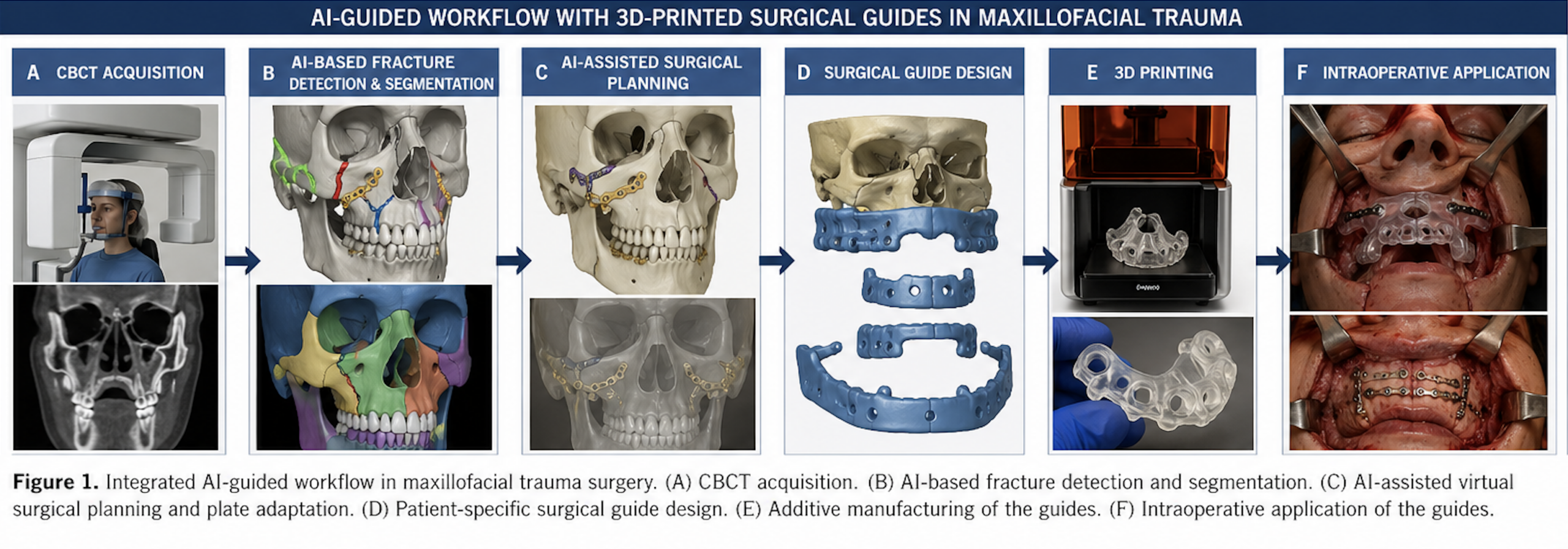
2.4 Intervention: AI-Guided Workflow
The intervention group underwent a fully integrated AI-assisted workflow consisting of:
- Automated fracture detection using CBCT imaging
- AI-based segmentation and anatomical reconstruction
- AI-assisted virtual surgical planning
- Design of patient-specific surgical guides
- Additive manufacturing using medical-grade polymer materials
Patient-specific guides were fabricated based on the AI-generated surgical plan and used intraoperatively to assist fracture reduction and fixation.
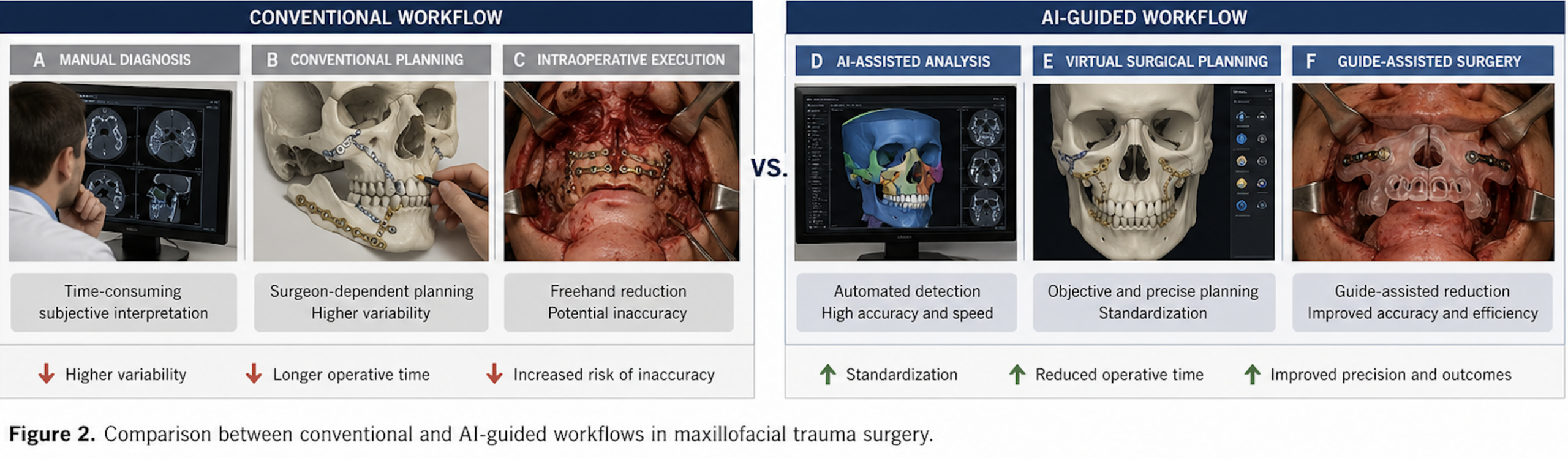
2.5 Control Group: Conventional Workflow
Patients in the control group received standard surgical care, including:
- Manual interpretation of CBCT imaging
- Surgeon-dependent planning
- Intraoperative freehand fracture reduction
No AI assistance or patient-specific guides were used.
2.6 Surgical Procedure
All procedures were performed under general anesthesia by experienced maxillofacial surgeons.
In the intervention group, patient-specific guides were positioned intraoperatively to assist reduction and fixation. In the control group, fracture reduction was performed manually based on surgeon experience.
Postoperative CBCT imaging was obtained in all patients to assess surgical accuracy.
The integrated AI-guided workflow, including CBCT-based fracture detection, segmentation, surgical planning, and patient-specific guide fabrication, is illustrated in Figure 1.
2.7 Outcome Measures
Primary endpoint:
- Surgical accuracy, defined as the deviation (mm) between planned and postoperative fracture alignment
Secondary endpoints:
- Operative time (minutes)
- Complication rate (intraoperative and postoperative)
- Workflow efficiency and intraoperative predictability
2.8 Statistical Analysis
Statistical analysis was performed using standard statistical software.
- Continuous variables were expressed as mean ± standard deviation
- Group comparisons were performed using independent t-tests
- Categorical variables were analyzed using chi-square tests
- A p-value < 0.05 was considered statistically significant
3. RESULTS
3.1 Patient Characteristics
A total of 104 patients were randomized equally into two groups (n = 52 per group). Demographic characteristics, including age, sex distribution, and fracture patterns, were comparable between the intervention and control groups, with no statistically significant differences.
A total of 104 patients were randomized into the intervention and control groups. Baseline demographic and clinical characteristics were comparable between both groups (Table 1).
| Step | AI Component | 3D Printing Component | Clinical Benefit |
| Detection | Automated fracture detection | — | Faster diagnosis |
| Segmentation | AI-based modeling | — | Precision |
| Planning | Decision support | — | Standardization |
| Guide Design | AI optimization | Patient-specific guides | Accuracy |
| Surgery | Intraoperative support | Guides | Reduced time |
| Outcome | Prediction models | — | Improved follow-up |
Table 1. Integrated AI and additive manufacturing workflow in maxillofacial trauma surgery
3.2 Surgical Accuracy
Postoperative CBCT analysis demonstrated significantly improved accuracy in the AI-guided group.
- Intervention group: 1.1 ± 0.5 mm
- Control group: 2.3 ± 0.9 mm
This difference was statistically significant (p < 0.001), indicating superior precision of AI-guided surgical execution.
3.3 Operative Time
The use of AI-guided 3D-printed surgical guides resulted in a significant reduction in operative time:
- Intervention group: 75 ± 14 minutes
- Control group: 105 ± 20 minutes
This corresponds to a 29% reduction in operative time (p < 0.001).
3.4 Complications
Complication rates were lower in the intervention group, although not statistically significant:
- Intervention group: 2 cases (3.8%)
- Control group: 5 cases (9.6%)
No major complications or adverse events related to the AI workflow or surgical guides were observed.
3.5 Workflow Efficiency
The AI-guided workflow demonstrated:
- reduced inter-surgeon variability
- improved intraoperative predictability
- enhanced workflow standardization
Surgeons reported increased confidence during fracture reduction when using patient-specific guides.
4. DISCUSSION
This randomized controlled trial provides high-level clinical evidence supporting the integration of artificial intelligence and additive manufacturing in maxillofacial trauma surgery. While previous studies have demonstrated the diagnostic capabilities of AI systems in fracture detection and planning [1–8], the present study extends these findings into a controlled clinical setting, directly comparing AI-guided workflows with conventional surgical approaches.
Compared to conventional workflows, the AI-guided approach demonstrates improved efficiency, accuracy, and standardization (Figure 2).
The significant improvement in surgical accuracy observed in this study is consistent with previous reports on computer-assisted maxillofacial surgery. Virtual surgical planning and CAD/CAM-based techniques have demonstrated accuracy ranges between 1.0 and 2.5 mm [9–11]. The present findings indicate that AI-guided workflows not only achieve comparable precision but may further reduce variability associated with manual planning and intraoperative decision-making.
A key advantage of the proposed approach is the translation of digital planning into physical execution through patient-specific 3D-printed guides. Previous literature has highlighted the potential of additive manufacturing to improve surgical outcomes in craniofacial reconstruction [12,13]. However, these studies often relied on manual or semi-automated planning processes. By integrating AI-based analysis, the present workflow represents a fully automated pipeline from imaging to surgical execution.
The observed reduction in operative time (29%) represents a clinically relevant improvement. Reduced operative time has been associated with lower perioperative risk, decreased anesthesia exposure, and improved operating room efficiency [14]. In addition, workflow standardization may reduce surgeon-dependent variability and improve reproducibility across institutions.
From a broader perspective, the integration of AI and additive manufacturing reflects a paradigm shift toward personalized and precision surgery. Similar developments have been reported in other surgical disciplines, including orthopedics and neurosurgery, where AI-assisted planning and patient-specific instrumentation have improved outcomes and reproducibility [15,16]. The present study confirms that these concepts are transferable to maxillofacial trauma surgery.
Despite these strengths, several limitations must be acknowledged. First, the sample size, although adequate for detecting differences in primary endpoints, may limit generalizability. Second, the study was conducted in specialized centers with access to advanced technology, which may not be universally available. Third, long-term outcomes were not assessed, and further studies are required to evaluate functional and aesthetic results over time.
Future research should focus on multicenter randomized trials with larger patient populations, as well as cost-effectiveness analyses and integration of predictive AI models for postoperative outcomes. Additionally, the incorporation of real-time intraoperative navigation and augmented reality may further enhance the clinical utility of AI-guided workflows.
In conclusion, this study demonstrates that AI-guided 3D-printed surgical guides significantly improve surgical accuracy and efficiency compared to conventional workflows. These findings support the clinical adoption of integrated AI-driven systems and represent a major step toward fully personalized maxillofacial trauma surgery.
5. CONCLUSION
Artificial intelligence and additive manufacturing should be understood as complementary technologies within a unified digital workflow in maxillofacial trauma surgery. Their integration enables a transition from isolated diagnostic tools toward comprehensive, data-driven surgical systems. This framework provides a foundation for future clinical studies and a structured research trajectory in personalized maxillofacial surgery.
6. ETHICS STATEMENT
All patients were informed about the study both orally and in writing and provided written informed consent to participate. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Hochschule Zurich, in Zurich, Switzerland.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Yildirim A, Hertach R, Yildirim V. Artificial intelligence–assisted detection of maxillofacial fractures on digital volume tomography. J Med Dent. 2026;2:44–52.
[2] Yildirim A, Hertach R, Yildirim V. External multicenter validation of an AI system for CBCT-based fracture detection. J Med Dent. 2026;2:70–81.
[3] Yildirim A, Hertach R, Yildirim V. AI-assisted clinical decision support in maxillofacial trauma. J Med Dent. 2026;2:82–92.
[4] Yildirim A, Hertach R, Yildirim V. AI-assisted surgical planning in midfacial fractures. J Med Dent. 2026;2:100–108.
[5] Yildirim A, Hertach R, Yildirim V. AI-based outcome prediction in maxillofacial trauma. J Med Dent. 2026;2:109–117.
[6] Liu Z, Tang Z, Wang H, et al. Deep learning–based automatic segmentation of maxillofacial fractures on CBCT images. Int J Comput Assist Radiol Surg. 2021;16(7):1235–1244.
[7] Martelli N, Serrano C, van den Brink H, Pineau J, Prognon P, Borget I, El Batti S. Advantages and disadvantages of 3-dimensional printing in surgery: a systematic review. Surgery. 2016;159(6):1485–1500.
[8] Rengier F, Mehndiratta A, von Tengg-Kobligk H, et al. 3D printing based on imaging data: review of medical applications. Int J Comput Assist Radiol Surg. 2010;5(4):335–341.
[9] Roser SM, Ramachandra S, Blair H, et al. Accuracy of virtual surgical planning in mandibular reconstruction. J Oral Maxillofac Surg. 2010;68(11):2824–2832.
[10] Xia JJ, Gateno J, Teichgraeber JF. New protocol for craniomaxillofacial surgical planning. J Oral Maxillofac Surg. 2009;67(10):2093–2106.
[11] Foley BD, Thayer WP, Honeybrook A, et al. CAD/CAM in mandibular reconstruction. J Oral Maxillofac Surg. 2013;71(2):e111–e119.
[12] Mitsouras D, Liacouras P, Imanzadeh A, et al. Medical 3D printing for radiology. Radiographics. 2015;35(7):1965–1988.
[13] Wong KC. 3D printing in orthopedics. Orthop Res Rev. 2016;8:57–66.
[14] Probst FA, Metzler P, Ehrenfeld M, Cornelius CP. Computer-assisted mandibular reconstruction. J Craniomaxillofac Surg. 2016;44(9):1415–1421.
[15] Topol EJ. High-performance medicine. Nat Med. 2019;25(1):44–56.
[16] Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification with deep neural networks. Nature. 2017;542(7639):115–118.
