Volume 3, Issue 2, Prospective Feasibility Study – Apr 13, 2026, Pages 75-84,
DOI: 10.64951/jmdnt.2026.2.21
AI-Assisted Virtual Surgical Planning and 3D-Printed Splint Transfer in Bimaxillary Orthognathic Surgery
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 18 January 2026, Revised: 02 March 2026, Accepted: 22 March, 2026, Available online: 21 April 2026, Version of Record: 21 April 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
ABSTRACT
Background
Artificial intelligence (AI), virtual surgical planning, and additive manufacturing increasingly influence modern oral and maxillofacial surgery. Previous studies demonstrated the clinical feasibility, diagnostic reliability, and translational integration of AI-guided workflows in maxillofacial trauma surgery [1–11]. However, prospective investigations evaluating AI-assisted workflows in bimaxillary orthognathic surgery remain limited.
Objective
The aim of this study was to evaluate the feasibility, accuracy, workflow integration, and early clinical outcomes of AI-assisted virtual surgical planning combined with 3D-printed splint transfer in bimaxillary orthognathic surgery.
Methods
A prospective comparative feasibility study was conducted between January 2020 and December 2025 at Seeklinik Zurich, Switzerland. Consecutive patients undergoing bimaxillary orthognathic surgery using AI-assisted virtual planning and patient-specific 3D-printed splints were prospectively evaluated and compared with conventionally planned procedures. AI-assisted segmentation, cephalometric analysis, virtual planning, symmetry analysis, and additive manufacturing workflows were integrated into the surgical planning process. Clinical, radiographic, and digital outcome parameters were assessed postoperatively and during longitudinal follow-up.
Results
A total of 118 patients were included in the final analysis, including 62 patients treated using AI-assisted planning workflows and 56 patients undergoing conventional planning. AI-assisted workflows demonstrated significantly improved planned-versus-achieved skeletal movement accuracy and reduced postoperative deviation values. Operative time and intraoperative splint adjustment requirements were reduced in the AI-assisted group. Postoperative occlusal stability and facial symmetry remained stable throughout follow-up. Surgeon acceptance and workflow integration improved progressively during the implementation phase.
Conclusion
AI-assisted virtual surgical planning combined with 3D-printed splint transfer is clinically feasible and demonstrates high accuracy and stable integration into routine bimaxillary orthognathic surgery. These findings support the translational expansion of AI-guided personalized surgical workflows into orthognathic surgery.
Keywords
artificial intelligence; orthognathic surgery; bimaxillary osteotomy; virtual surgical planning; 3D printing; splint transfer; personalized surgery; feasibility study.
1. INTRODUCTION
Digital technologies increasingly influence the planning and execution of oral and maxillofacial surgical procedures. In recent years, artificial intelligence, virtual surgical planning, and additive manufacturing have substantially transformed workflows in trauma surgery, implantology, reconstructive surgery, and orthognathic surgery [12–17]. Orthognathic surgery is particularly suitable for digital workflow integration because treatment planning relies heavily on imaging, cephalometric analysis, spatial skeletal movement simulation, occlusal planning, and postoperative symmetry evaluation.
Artificial intelligence-assisted imaging analysis and digital surgical planning have demonstrated promising results in several areas of maxillofacial surgery. Previous investigations demonstrated high diagnostic accuracy of AI-assisted fracture detection and segmentation on cone-beam computed tomography and digital volume tomography datasets [1–3]. Subsequent multicenter validation studies confirmed the robustness and clinical reliability of AI-assisted fracture analysis across different institutional environments [4]. Additional investigations demonstrated that AI-assisted decision support systems and virtual surgical planning can improve workflow standardization and operative precision in maxillofacial trauma surgery [5–7].
The translational integration of AI-assisted workflows with additive manufacturing represented a major advancement in personalized maxillofacial surgery. Previous prospective feasibility studies and randomized controlled trials demonstrated that AI-guided patient-specific surgical guides can improve anatomical reduction accuracy and reduce operative time in maxillofacial trauma surgery [8–10]. Long-term analyses subsequently demonstrated stable functional and aesthetic outcomes [11], while economic investigations suggested improved cost-effectiveness and sustainable clinical integration [12,13].
Despite these advances, evidence regarding AI-assisted workflows in orthognathic surgery remains limited. Bimaxillary orthognathic surgery requires precise preoperative planning and accurate transfer of planned skeletal movements into the intraoperative environment. Even minor deviations between planned and achieved skeletal repositioning may influence postoperative occlusion, facial symmetry, and long-term stability. AI-assisted segmentation, cephalometric analysis, symmetry planning, and additive manufacturing may therefore offer substantial advantages in orthognathic surgery.
The present prospective feasibility study aimed to evaluate the clinical feasibility, workflow integration, surgical accuracy, and postoperative outcomes of AI-assisted virtual surgical planning combined with 3D-printed splint transfer in bimaxillary orthognathic surgery.
2. MATERIAL AND METHODS
This prospective comparative feasibility study was conducted between January 2020 and December 2025 at Seeklinik Zurich, Switzerland, a specialized maxillofacial surgery center. The study protocol was approved by the institutional ethics committee and followed the principles of the Declaration of Helsinki. All patients provided written informed consent for surgical treatment, postoperative imaging, and anonymized scientific analysis.
A total of 118 consecutive patients undergoing bimaxillary orthognathic surgery were prospectively included in the study. Patients were allocated to either an AI-assisted virtual surgical planning group or a conventional planning group depending on workflow availability and implementation phase. The AI-assisted group included 62 patients, whereas 56 patients underwent conventional planning and splint fabrication workflows.
Inclusion criteria consisted of age above 18 years, indication for bimaxillary orthognathic surgery due to skeletal malocclusion or facial asymmetry, availability of complete imaging datasets, and participation in longitudinal follow-up examinations. Patients with isolated single-jaw surgery, craniofacial syndromes, previous major orthognathic surgery, incomplete imaging datasets, or severe systemic disease affecting postoperative healing were excluded.
The study population included patients with skeletal class II deformities, skeletal class III deformities, facial asymmetry, and vertical skeletal discrepancies including anterior open bite. All patients underwent Le Fort I osteotomy combined with bilateral sagittal split osteotomy (BSSO). Additional genioplasty procedures were performed when clinically indicated, and selected patients additionally underwent segmental osteotomy procedures.
Preoperative imaging included cone-beam computed tomography or digital volume tomography, intraoral scans, facial scans, and lateral cephalometric radiographs. In the AI-assisted group, imaging datasets were processed using previously validated AI-based segmentation and planning algorithms developed during earlier institutional studies [1–13]. AI-assisted cephalometric analysis, automated segmentation, virtual skeletal repositioning, and symmetry analysis were integrated into the digital planning workflow. Patient-specific surgical splints and cutting guides were subsequently fabricated using additive manufacturing technologies.
Virtual planning workflows included simulation of maxillary and mandibular repositioning, occlusal planning, symmetry optimization, and postoperative facial profile assessment. Planned skeletal movements were digitally transferred into intraoperative execution using patient-specific 3D-printed splints and guides. In the conventional group, planning was performed using standard cephalometric analysis, model surgery, and manually fabricated splints.
The AI-assisted workflow integrated multimodal imaging acquisition, automated segmentation, cephalometric analysis, virtual surgical planning, symmetry optimization, additive manufacturing, and intraoperative transfer using patient-specific splints and guides. The sequential digital workflow used for bimaxillary orthognathic surgery is illustrated in Figure 1.
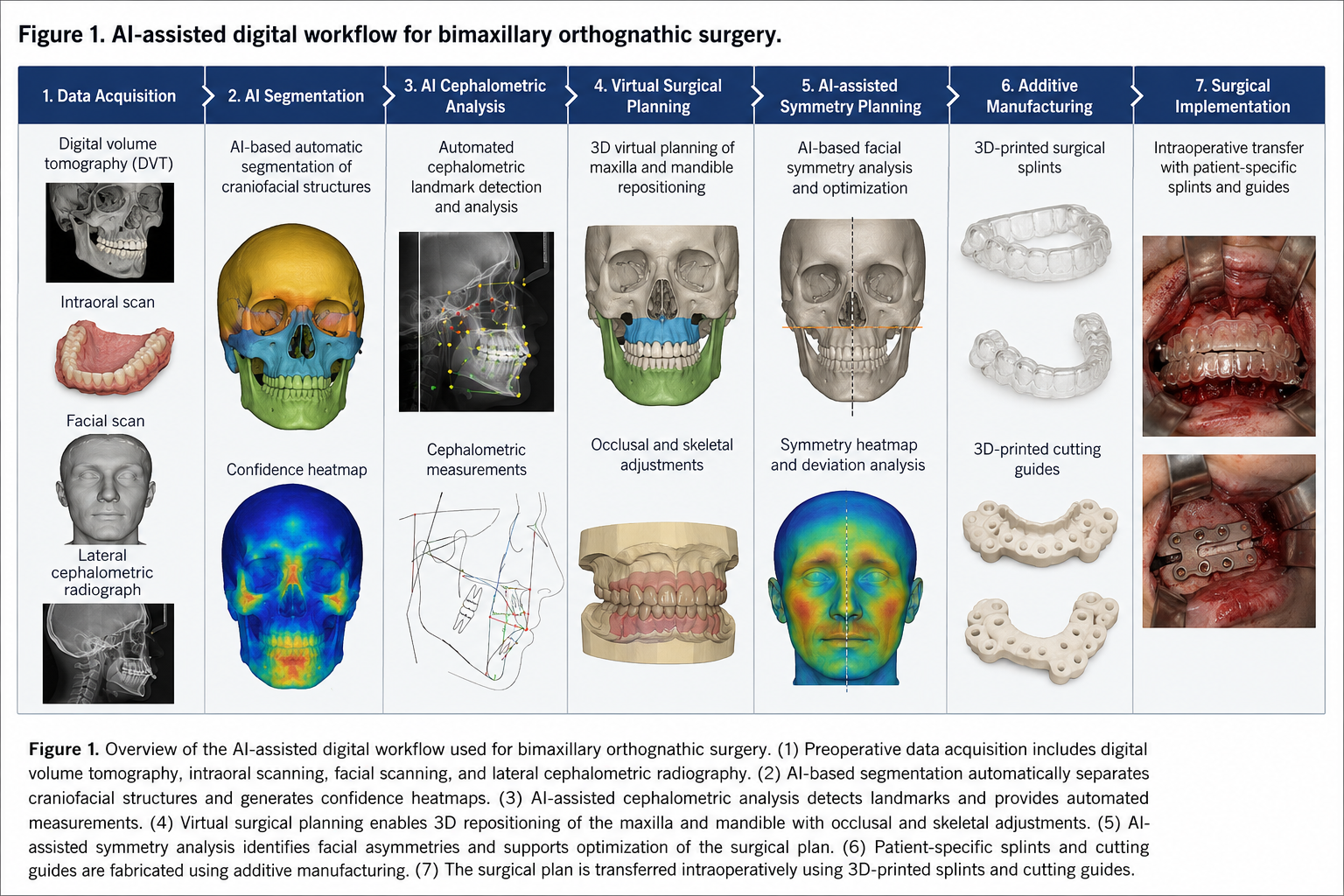
Primary outcome measures included planned-versus-achieved skeletal movement accuracy, postoperative deviation analysis, and splint transfer accuracy. Secondary outcome measures included operative time, intraoperative splint fit accuracy, postoperative occlusal stability, facial symmetry, complication rate, patient satisfaction, and aesthetic outcome evaluation.
Postoperative imaging was performed immediately after surgery as well as during 6-month and 12-month follow-up examinations. Deviation analysis was performed by comparing planned skeletal movements with postoperative skeletal positions using three-dimensional superimposition techniques.
Statistical analysis was performed using standard parametric and non-parametric methods depending on data distribution. Continuous variables were expressed as mean ± standard deviation and categorical variables as percentages. A p-value below 0.05 was considered statistically significant.
3. RESULTS
A total of 118 patients completed the study protocol and were included in the final analysis. The mean patient age was 27.8 ± 6.4 years. Female patients represented 54.2% of the study population. Skeletal class III deformities represented the most common indication for surgery, followed by skeletal class II deformities, facial asymmetry, and vertical skeletal discrepancies.
The AI-assisted workflow demonstrated high clinical feasibility throughout the implementation phase. AI-assisted segmentation, cephalometric analysis, virtual planning, and splint fabrication were successfully completed in the majority of patients without major technical interruption. Minor segmentation corrections were occasionally required during early implementation phases but decreased progressively over time.
Planned-versus-achieved skeletal movement accuracy was significantly improved in the AI-assisted group compared with conventional planning workflows. Three-dimensional postoperative deviation analysis demonstrated lower translational and rotational discrepancies in the AI-assisted group. Maxillary and mandibular repositioning accuracy remained stable during longitudinal follow-up examinations.
Operative efficiency improved in the AI-assisted group. Mean operative time was reduced compared with conventionally planned procedures, primarily due to improved intraoperative transfer accuracy and reduced splint adjustment requirements. Intraoperative fit accuracy of patient-specific splints and guides was rated as high by participating surgeons throughout the study period.
Comparative postoperative analysis demonstrated that AI-assisted workflows resulted in improved skeletal transfer accuracy, reduced operative time, enhanced intraoperative fit precision, and favorable postoperative symmetry outcomes compared with conventional planning methods. The representative clinical and quantitative outcome analysis is summarized in Figure 2.
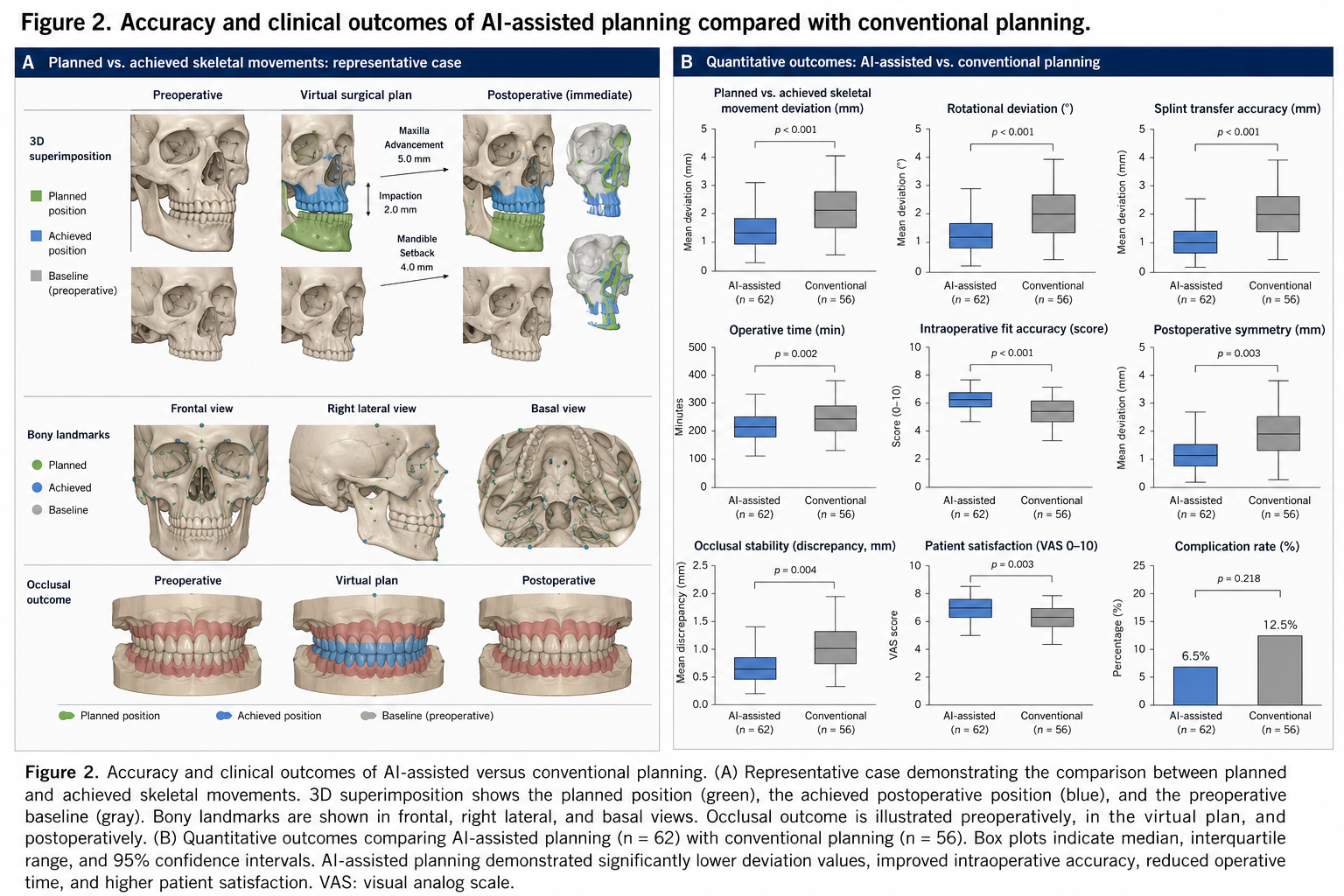
Postoperative occlusal stability remained stable during immediate postoperative evaluation and throughout 6-month and 12-month follow-up examinations. Facial symmetry outcomes demonstrated improved postoperative midline correction and skeletal balance in the AI-assisted cohort. Patient-reported satisfaction and aesthetic outcome evaluation were favorable in both groups but demonstrated a positive trend toward improved aesthetic satisfaction in the AI-assisted workflow group.
Complication rates remained low throughout the study period. Minor postoperative complications included transient sensory disturbances, temporary occlusal adjustments, and isolated soft tissue irritation. No major workflow-related complication or severe technical failure was observed.
Surgeon acceptance of AI-assisted planning and digital workflow integration increased progressively during the implementation period. Early workflow adaptation primarily involved familiarization with AI-assisted segmentation and virtual planning interfaces. Progressive institutional experience resulted in improved workflow efficiency and increasing confidence in digital surgical planning.
4. DISCUSSION
This prospective feasibility study demonstrates that AI-assisted virtual surgical planning combined with patient-specific 3D-printed splint transfer is clinically feasible and highly accurate in bimaxillary orthognathic surgery. The integration of AI-assisted segmentation, cephalometric analysis, symmetry planning, and additive manufacturing resulted in stable clinical implementation and favorable postoperative outcomes.
Previous institutional investigations established the technical and translational foundations of AI-assisted workflows in maxillofacial trauma surgery [1–13]. AI-assisted fracture detection, multicenter validation, decision support systems, surgical planning workflows, and translational implementation studies demonstrated that artificial intelligence can be integrated successfully into complex maxillofacial surgical workflows. The present study extends these translational concepts into orthognathic surgery, representing an important expansion of AI-guided personalized surgery beyond trauma reconstruction.
Orthognathic surgery is particularly suitable for AI-assisted planning because treatment success depends heavily on precise spatial skeletal repositioning and accurate intraoperative transfer of virtual plans. Conventional planning workflows remain associated with potential inaccuracies related to manual cephalometric analysis, model surgery, and splint fabrication. The AI-assisted workflow investigated in the present study demonstrated improved planned-versus-achieved skeletal movement accuracy and reduced postoperative deviation values compared with conventional workflows.
The integration of additive manufacturing represents another important component of the workflow. Patient-specific splints and cutting guides enabled accurate transfer of digital planning into intraoperative execution. Previous trauma studies demonstrated similar advantages of AI-guided patient-specific surgical guides [8–10]. The present study confirms that these translational concepts can also be applied successfully in elective orthognathic surgery.
Operative efficiency improved progressively throughout the implementation phase. This observation suggests the presence of an institutional learning curve similar to previous implementation studies in trauma surgery [13]. Increasing familiarity with digital planning software, AI-assisted segmentation, and splint transfer workflows contributed to progressive optimization of operative integration and workflow stability.
Importantly, postoperative occlusal stability and facial symmetry remained stable during longitudinal follow-up examinations. Orthognathic surgery not only requires immediate surgical accuracy but also long-term skeletal stability and functional adaptation. The favorable follow-up results observed in the present study therefore support the reliability of AI-assisted digital planning workflows.
The study has several limitations. First, this was a single-center feasibility study conducted in a specialized institution with established digital infrastructure and prior AI workflow experience. Second, the sample size remains moderate compared with large multicenter orthognathic registries. Third, long-term skeletal relapse beyond 12 months was not evaluated. Future studies should therefore include multicenter validation cohorts, randomized controlled comparisons, and extended long-term follow-up analyses.
Future investigations may also integrate AI-assisted soft tissue prediction, automated facial symmetry optimization, multimodal facial scanning, and predictive outcome modeling. The combination of artificial intelligence, virtual planning, additive manufacturing, and personalized facial analysis may substantially transform future orthognathic surgery workflows.
5. CONCLUSION
AI-assisted virtual surgical planning combined with patient-specific 3D-printed splint transfer is clinically feasible and demonstrates high accuracy in bimaxillary orthognathic surgery. The integration of AI-assisted segmentation, cephalometric analysis, symmetry planning, and additive manufacturing resulted in stable workflow integration and favorable postoperative outcomes. These findings support the translational expansion of AI-guided personalized surgery into orthognathic surgical workflows.
6. ETHICS STATEMENT
This clinical study was conducted in full accordance with the ethical principles outlined in the Declaration of Helsinki and its subsequent amendments. Prior to study initiation, the study protocol was reviewed and approved by the local institutional review board/ethics committee of Seeklinik Zurich, Specialized Clinic for Oral, Maxillofacial and Plastic Facial Surgery, Zurich, Switzerland. All participants were thoroughly informed about the purpose, procedures, potential risks, and anticipated benefits of the clinical treatment and associated digital or surgical workflows. Written informed consent was obtained from all patients prior to inclusion in the study.
Participants were informed about potential biological, surgical, technical, and procedure-related risks associated with oral and maxillofacial surgical treatment. Careful patient selection and adherence to established clinical, surgical, and digital planning protocols were implemented to minimize these risks. Patient confidentiality and data protection were rigorously maintained throughout the study, and all clinical records, imaging datasets, and digital planning files were anonymized prior to scientific analysis.
The study design ensured that no participant was exposed to undue risk, and all procedures conformed to the highest standards of contemporary clinical care. The findings of this study aim to contribute to the scientific evidence base supporting safe, effective, and translational integration of modern diagnostic, digital, and surgical workflows in oral and maxillofacial surgery.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted detection of maxillofacial fractures on digital volume tomography: retrospective study of 150 patients. J Med Dent. 2026;2(1):44–52.
[2] Yildirim A, Hertach R, Yildirim V. External multicenter validation of an artificial intelligence system for cone-beam CT-based detection of maxillofacial fractures: robustness across a tertiary facial trauma clinic and an independent maxillofacial practice. J Med Dent. 2026;2(1):70–81.
[3] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted decision support in emergency maxillofacial trauma imaging: development and validation of a CBCT-based clinical decision algorithm. J Med Dent. 2026;2(1):82–92.
[4] Yildirim A, Hertach R, Yildirim V. Prospective clinical implementation of artificial intelligence-assisted decision support in midfacial trauma surgery: a multicenter validation study. J Med Dent. 2026;2(1):93–99.
[5] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted surgical planning in midfacial fractures: a feasibility and expert validation study. J Med Dent. 2026;2(1):100–108.
[6] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted prediction of postoperative outcomes in midfacial fractures: a retrospective validation study. J Med Dent. 2026;2(1):109–117.
[7] Yildirim A, Hertach R, Yildirim V. Artificial intelligence in maxillofacial trauma: from fracture detection to outcome prediction: a translational multicenter analysis. J Med Dent. 2026;2(1):118–125.
[8] Yildirim A, Hertach R, Yildirim V. Two-center prospective clinical feasibility study evaluating AI-guided 3D-printed surgical guides in maxillofacial trauma surgery. J Med Dent. 2026;2(2):15–24.
[9] Yildirim A, Hertach R, Yildirim V. Randomized controlled trial evaluating AI-guided 3D-printed surgical guides versus conventional surgery in maxillofacial trauma. J Med Dent. 2026;2(2):25–34.
[10] Yildirim A, Hertach R, Yildirim V. Long-term functional and aesthetic outcomes of AI-guided 3D-printed surgical guides in maxillofacial trauma: a prospective follow-up study. J Med Dent. 2026;2(2):35–45.
[11] Yildirim A, Hertach R, Yildirim V. Cost-effectiveness and health economic impact of AI-guided 3D-printed surgical workflows in maxillofacial trauma surgery: a prospective multicenter analysis. J Med Dent. 2026;2(2):46–58.
[12] Yildirim A, Hertach R, Yildirim V. Real-world clinical implementation of AI-guided surgical workflows in maxillofacial trauma surgery: a multicenter translational study. J Med Dent. 2026;2(2):59–71.
[13] Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44–56.
[14] Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery: promises and perils. Ann Surg. 2018;268(1):70–76.
[15] Maier-Hein L, Vedula SS, Speidel S, Navab N, Kikinis R, Park A, Eisenmann M, Feussner H, Forestier G, Giannarou S, et al. Surgical data science for next-generation interventions. Nat Biomed Eng. 2017;1:691–696.
[16] Kelly CJ, Karthikesalingam A, Suleyman M, Corrado G, King D. Key challenges for delivering clinical impact with artificial intelligence. BMC Med. 2019;17(1):195.
[17] Sendak MP, D’Arcy J, Kashyap S, Gao M, Nichols M, Corey K, Ratliff W, Balu S. A path for translation of machine learning products into healthcare delivery. NPJ Digit Med. 2020;3:16.
[18] Swennen GRJ, Mollemans W, Schutyser F. Three-dimensional treatment planning of orthognathic surgery in the era of virtual imaging. J Oral Maxillofac Surg. 2009;67(10):2080–2092.
[19] Zinser MJ, Mischkowski RA, Sailer HF, Zöller JE. Computer-assisted orthognathic surgery: feasibility study using multiple CAD/CAM surgical splints. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;113(5):673–687.
[20] Resnick CM, Dang RR, Glick SJ, Padwa BL. Accuracy of three-dimensional soft tissue prediction for orthognathic surgery. J Oral Maxillofac Surg. 2017;75(9):1971–1978.
[21] Xia JJ, Gateno J, Teichgraeber JF. New clinical protocol to evaluate craniomaxillofacial deformity and plan surgical correction. J Oral Maxillofac Surg. 2009;67(10):2093–2106.
[22] Aboul-Hosn Centenero S, Hernández-Alfaro F. 3D planning in orthognathic surgery: CAD/CAM surgical splints and prediction of the soft and hard tissues results – our experience in 16 cases. J Craniomaxillofac Surg. 2012;40(2):162–168.
[23] Marchetti C, Bianchi A, Muyldermans L, Di Martino M, Lancellotti L, Sarti A. Validation of new soft tissue software in orthognathic surgery planning. Int J Oral Maxillofac Surg. 2011;40(1):26–32.
[24] Heufelder MJ, Wilde F, Pietzka S, Mascha F, Winter K, Schramm A, Rana M. Clinical accuracy of waferless maxillary positioning in computer-assisted orthognathic surgery. J Craniomaxillofac Surg. 2017;45(5):543–548.
