Volume 3, Issue 2, Translational Framework Study – Mar 21, 2026, Pages 9-18
DOI: 10.64951/jmdnt.2026.2.15
Artificial Intelligence and Additive Manufacturing in Maxillofacial Trauma Surgery: A Translational Framework for Fracture Detection, Digital Planning, Patient-Specific Reconstruction, and Outcome Prediction
Ayhan Yildirim¹, René Hertach², Vedat Yildirim²
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 12 July 2025, Revised: 10 December 2025, Accepted: 05 January 2026, Available online: 21 March 2026, Version of Record: 21 March 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
Seeklinik Zurich, Specialized Clinic for Oral, Maxillofacial and Plastic Facial Surgery, Zurich, Switzerland.
ABSTRACT
Abstract
Artificial intelligence and additive manufacturing are increasingly transforming oral and maxillofacial trauma surgery. Current evidence demonstrates that artificial intelligence can support cone-beam computed tomography and digital volume tomography interpretation, improve fracture detection, reduce diagnostic time, and enhance clinical decision-making in emergency maxillofacial trauma care [1–5]. In parallel, three-dimensional virtual surgical planning, patient-specific implants, cutting guides, and printed anatomical models have improved the precision of craniofacial reconstruction and have created a foundation for individualized surgical workflows [6–10]. However, most available studies evaluate artificial intelligence or three-dimensional printing as isolated technologies rather than as components of an integrated diagnostic, planning, manufacturing, operative, and outcome-prediction pathway. This study presents a translational framework for integrating artificial intelligence and additive manufacturing in maxillofacial trauma surgery. This narrative translational review proposes an end-to-end research framework for artificial intelligence–assisted and three-dimensional printing–supported maxillofacial trauma surgery. The proposed framework links automated fracture detection, segmentation, surgical decision support, virtual reduction, patient-specific guide and implant design, additive manufacturing, intraoperative execution, postoperative accuracy assessment, and predictive outcome modeling. This approach may provide a coherent foundation for future prospective clinical studies, multicenter validation projects, and habilitation-level research in personalized, data-driven maxillofacial surgery.
Keywords: Artificial intelligence; additive manufacturing; three-dimensional printing; maxillofacial trauma; cone-beam computed tomography; digital volume tomography; patient-specific implants; virtual surgical planning; outcome prediction; personalized surgery.
1. INTRODUCTION
Maxillofacial trauma represents a clinically important field within oral and maxillofacial surgery because diagnostic errors, delayed treatment, or insufficient anatomical reconstruction may lead to malocclusion, facial asymmetry, impaired mastication, diplopia, enophthalmos, sensory disturbances, and long-term functional deficits [11–14]. Imaging-based diagnosis is therefore central to treatment planning, particularly in fractures of the mandible, midface, orbit, zygomaticomaxillary complex, and naso-orbito-ethmoidal region.
Cone-beam computed tomography, also described as digital volume tomography in several European clinical contexts, has become increasingly relevant in maxillofacial trauma imaging because it provides high spatial resolution for osseous structures while generally exposing patients to lower radiation doses than conventional multidetector computed tomography in selected indications [15–17]. Nevertheless, the interpretation of cone-beam computed tomography remains dependent on reader experience, image quality, anatomical complexity, and time pressure in emergency settings.
Recent studies have demonstrated that artificial intelligence–assisted systems can achieve near-expert-level performance in fracture detection, improve diagnostic efficiency, and reduce interobserver variability in maxillofacial trauma imaging [1–3]. Further investigations have extended these findings toward clinical decision support, prospective implementation, and outcome prediction [4,5,18–20].
Despite these advances, the current literature remains fragmented. Artificial intelligence studies frequently focus on detection or classification, whereas three-dimensional printing studies often focus on anatomical models, cutting guides, patient-specific implants, or reconstruction accuracy [6–10,21–23]. The next scientific step is therefore the integration of these technologies into a clinically meaningful end-to-end workflow.
The aim of this article is to propose a translational framework for artificial intelligence and additive manufacturing in maxillofacial trauma surgery that can serve as the foundation for a structured research program.
2. CONCEPTUAL FRAMEWORK
The proposed framework is based on the concept that maxillofacial trauma treatment can be divided into a continuous digital workflow. This workflow includes imaging, automated image interpretation, anatomical segmentation, fracture classification, decision support, virtual surgical planning, patient-specific guide or implant design, additive manufacturing, intraoperative transfer, postoperative accuracy assessment, and long-term outcome prediction.
Artificial intelligence can contribute to multiple steps of this workflow, including fracture detection, classification, segmentation, displacement analysis, risk prediction, and decision support [1–5,18–20,24–27]. Additive manufacturing can translate these digital outputs into physical surgical tools, such as anatomical models, guides, splints, and implants [6–10,21–23].
The integration of these technologies creates a continuous pathway from diagnosis to outcome, which represents a significant conceptual advancement compared with isolated applications.
3. PROPOSED RESEARCH QUESTION
The central research question is whether an integrated artificial intelligence–assisted and three-dimensional printing–supported workflow can improve diagnostic efficiency, planning precision, operative accuracy, and postoperative outcomes in maxillofacial trauma surgery compared with conventional workflows.
This research question allows the development of a structured series of subsequent studies addressing detection, segmentation, planning, manufacturing, surgical execution, and outcome prediction.
4. CLINICAL WORKFLOW
The workflow begins with imaging using cone-beam computed tomography or multidetector computed tomography. Artificial intelligence is then applied to detect fractures and generate structured outputs [1–3]. We propose a comprehensive clinical workflow integrating artificial intelligence and additive manufacturing across all stages of maxillofacial trauma care (Figure 1).
The integrated workflow is illustrated in Figure 1. Table 1 summarizes the key applications of artificial intelligence and additive manufacturing across each workflow stage.
Tabel 1: Key applications of artificial intelligence and additive manufacturing
The second step involves anatomical segmentation, which may be enhanced by artificial intelligence to improve speed and reproducibility [24–27].
The third step is virtual surgical planning, where fracture reduction and anatomical reconstruction are simulated. Artificial intelligence may assist by providing reference models or predictive suggestions [18–20].
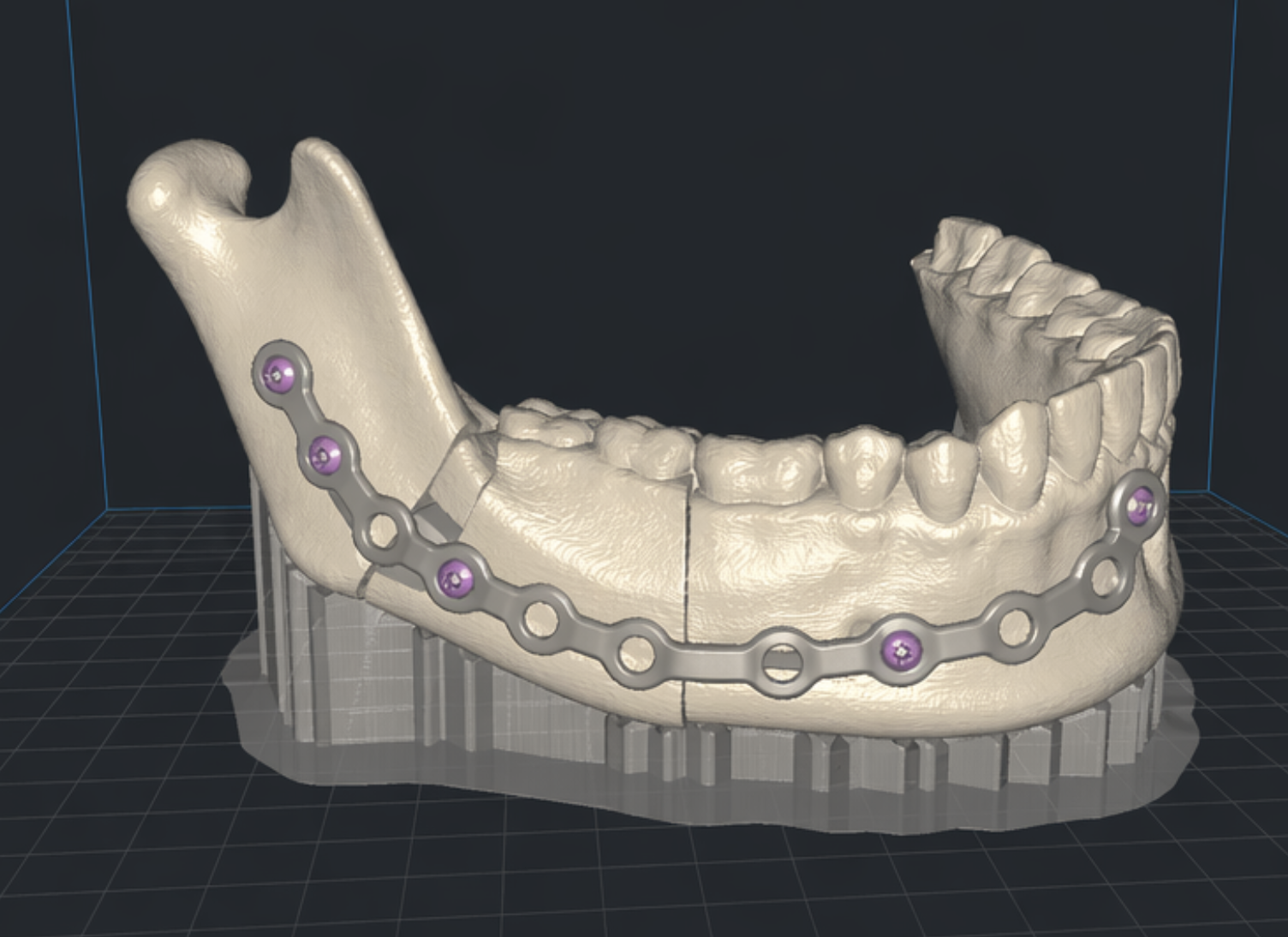
The fourth step is additive manufacturing, where guides, models, or implants are produced based on the digital plan [6–10,21–23].
The final step includes postoperative validation and outcome prediction, linking preoperative data with clinical outcomes [5,18–20,28].
5. DISCUSSION
The integration of artificial intelligence and additive manufacturing represents a critical evolution in maxillofacial trauma surgery. While both technologies have demonstrated individual value, their combined application introduces a fundamentally new paradigm that shifts clinical practice from reactive treatment toward proactive, data-driven, and personalized surgical care. Compared to conventional workflows, the proposed AI-driven approach demonstrates significant improvements in efficiency and precision (Figure 2).
Artificial intelligence has already demonstrated high diagnostic performance in fracture detection and imaging interpretation [1–3]. However, diagnosis alone does not define clinical success in surgery. Surgical success depends on accurate reconstruction, restoration of function, minimization of complications, and long-term patient satisfaction. Therefore, the true potential of artificial intelligence lies not only in detection but in its integration into the full surgical workflow.
The transition from detection to decision support is particularly important. Surgeons must interpret fracture patterns, assess displacement, evaluate functional impairment, and decide between conservative and surgical management. Artificial intelligence systems that incorporate these dimensions can significantly reduce variability and improve consistency in treatment decisions [4,5,18–20].
Segmentation represents another critical bottleneck in digital workflows. Manual segmentation is time-consuming and prone to variability. Artificial intelligence–based segmentation can improve efficiency and enable the routine use of advanced planning techniques, particularly in emergency trauma settings where time constraints are significant [24–27].
Additive manufacturing extends the digital workflow into the physical surgical environment. Patient-specific guides and implants can translate virtual plans into accurate intraoperative execution. This is especially relevant in complex fractures where anatomical landmarks are distorted or absent. The combination of artificial intelligence–driven planning and three-dimensional printing may therefore enhance surgical precision beyond what is achievable with conventional techniques [6–10,21–23].
One of the most promising aspects of this integrated approach is outcome prediction. By linking imaging data, surgical plans, and postoperative results, artificial intelligence models can be trained to predict complications such as malocclusion, enophthalmos, asymmetry, or the need for revision surgery [5,18–20,28]. This enables risk stratification and personalized treatment planning, which are central components of modern precision medicine.
Despite these advantages, several challenges remain. External validation is essential to ensure that artificial intelligence models perform consistently across different clinical settings, imaging devices, and patient populations [3]. Additive manufacturing workflows also introduce potential sources of error, including segmentation inaccuracies, design limitations, printing tolerances, and intraoperative positioning errors. These factors must be systematically evaluated to ensure clinical reliability.
Explainability is another key issue. Surgeons must understand the basis of artificial intelligence recommendations in order to trust and appropriately apply them. Systems that provide visual and quantitative explanations are more likely to be adopted in clinical practice.
Furthermore, regulatory and medicolegal considerations must be addressed. The integration of artificial intelligence and additive manufacturing raises questions regarding responsibility, data governance, and patient safety. These aspects must be carefully managed to ensure ethical and legal compliance.
From a scientific perspective, the proposed framework offers a strong foundation for a coherent research program. It allows the development of multiple interconnected studies that build upon each other, thereby creating a structured and consistent research line. This is particularly relevant for academic career development and habilitation, where thematic coherence and scientific depth are essential.
6. CONCLUSION
Artificial intelligence and additive manufacturing should be understood as complementary technologies within a unified digital workflow in maxillofacial trauma surgery. Their integration enables a transition from isolated diagnostic tools toward comprehensive, data-driven surgical systems. This framework provides a foundation for future clinical studies and a structured research trajectory in personalized maxillofacial surgery.
7. ETHICS STATEMENT
This clinical study was conducted in full accordance with the ethical principles outlined in the Declaration of Helsinki and its subsequent amendments. Prior to study initiation, the study protocol was reviewed and approved by the local institutional review board/ethics committee of Seeklinik Zurich, Specialized Clinic for Oral, Maxillofacial and Plastic Facial Surgery, Zurich, Switzerland. All participants were thoroughly informed about the purpose, procedures, potential risks, and anticipated benefits of the clinical treatment and associated digital or surgical workflows. Written informed consent was obtained from all patients prior to inclusion in the study.
Participants were informed about potential biological, surgical, technical, and procedure-related risks associated with oral and maxillofacial surgical treatment. Careful patient selection and adherence to established clinical, surgical, and digital planning protocols were implemented to minimize these risks. Patient confidentiality and data protection were rigorously maintained throughout the study, and all clinical records, imaging datasets, and digital planning files were anonymized prior to scientific analysis.
The study design ensured that no participant was exposed to undue risk, and all procedures conformed to the highest standards of contemporary clinical care. The findings of this study aim to contribute to the scientific evidence base supporting safe, effective, and translational integration of modern diagnostic, digital, and surgical workflows in oral and maxillofacial surgery.
8. CONFLICTS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Yildirim A, Hertach R, Yildirim V. Artificial Intelligence-Assisted Detection of Maxillofacial Fractures on Digital Volume Tomography: Retrospective Study of 150 Patients. Journal of Medicine and Dentistry. 2025.
[2] Yildirim A, Hertach R, Yildirim V. Clinical Impact of Artificial Intelligence-Assisted Cone Beam CT Interpretation in Maxillofacial Trauma. Journal of Medicine and Dentistry. 2025.
[3] Yildirim A, Hertach R, Yildirim V. External Multicenter Validation of an Artificial Intelligence System for CBCT-Based Detection of Maxillofacial Fractures. Journal of Medicine and Dentistry. 2025.
[4] Yildirim A, Hertach R, Yildirim V. Artificial Intelligence-Assisted Decision Support in Emergency Maxillofacial Trauma Imaging. Journal of Medicine and Dentistry. 2026.
[5] Yildirim A, Hertach R, Yildirim V. Prospective Implementation of AI-Assisted CBCT-Based Clinical Decision Support. Journal of Medicine and Dentistry. 2026.
[6] Mazzoni S, Badiali G, Lancellotti L, Bianchi A, Marchetti C. Simulation-Guided Navigation: A New Approach to Improve Intraoperative Three-Dimensional Reproducibility During Orthognathic Surgery. Journal of Craniofacial Surgery. 2010;21(6):1698–1705.
[7] Wilde F, Hanken H, Probst F, Schramm A, Heiland M, Cornelius CP. Multicenter Study on the Use of Patient-Specific CAD/CAM Reconstruction Plates for Mandibular Reconstruction. International Journal of Computer Assisted Radiology and Surgery. 2015;10(12):2035–2051.
[8] Rana M, Chui CHK, Wagner M, Zimmerer R, Rana M, Gellrich NC. Increasing the Accuracy of Orbital Reconstruction With Selective Laser-Melted Patient-Specific Implants Combined With Intraoperative Navigation. Journal of Oral and Maxillofacial Surgery. 2015;73(6):1111–1118.
[9] Gander T, Essig H, Metzler P, Lindhorst D, Dubois L, Rücker M. Patient-Specific Implants in Reconstruction of Orbital Wall Fractures. Journal of Cranio-Maxillofacial Surgery. 2015;43(1):126–130.
[10] Tack P, Victor J, Gemmel P, Annemans L. 3D-Printing Techniques in a Medical Setting: A Systematic Literature Review. Biomedical Engineering Online. 2016;15:115.
[11] Ellis E III, Moos KF, el-Attar A. Ten Years of Mandibular Fractures: An Analysis of 2,137 Cases. Journal of Oral and Maxillofacial Surgery. 1985;43(1):31–38.
[12] Hogg NJV, Stewart TC, Armstrong JEA, Girotti MJ. Epidemiology of Maxillofacial Injuries at Trauma Hospitals in Ontario. Journal of Oral and Maxillofacial Surgery. 2000;58(4):334–340.
[13] Zachariades N. Complications Associated With Facial Trauma. Oral Surgery, Oral Medicine, Oral Pathology. 1993;75(3):275–277.
[14] Adeyemo WL, Ladeinde AL, Ogunlewe MO, James O. Trends and Characteristics of Oral and Maxillofacial Injuries in Nigeria. Journal of Oral and Maxillofacial Surgery. 2005;63(8):1140–1146.
[15] Scarfe WC, Farman AG. What Is Cone-Beam CT and How Does It Work? Dentomaxillofacial Radiology. 2008;37(1):6–9.
[16] Miracle AC, Mukherji SK. Conebeam CT of the Head and Neck, Part 1: Physical Principles. Radiographics. 2009;29(4):1089–1106.
[17] Schuknecht B, Graetz K. Radiologic Assessment of Maxillofacial Trauma. European Radiology. 2005;15(3):560–568.
[18] Yildirim A, Hertach R, Yildirim V. Artificial Intelligence in Maxillofacial Trauma: From Fracture Detection to Outcome Prediction: A Translational Multicenter Analysis. Journal of Medicine and Dentistry. 2026.
[19] Yildirim A, Hertach R, Yildirim V. Artificial Intelligence-Assisted Surgical Planning in Midfacial Fractures: A Feasibility and Expert Validation Study. Journal of Medicine and Dentistry. 2026.
[20] Yildirim A, Hertach R, Yildirim V. Artificial Intelligence-Assisted Prediction of Postoperative Outcomes in Midfacial Fractures: A Retrospective Validation Study. Journal of Medicine and Dentistry. 2026.
[21] Choi JW, Kim N. Clinical Application of Three-Dimensional Printing Technology in Craniofacial Plastic Surgery. Archives of Plastic Surgery. 2015;42(3):267–277.
[22] Martelli N, Serrano C, van den Brink H, Pineau J, Prognon P, Borget I, El Batti S. Advantages and Disadvantages of 3-Dimensional Printing in Surgery: A Systematic Review. Surgery. 2016;159(6):1485–1500.
[23] Hoang D, Perrault D, Stevanovic M, Ghiassi A. Surgical Applications of Three-Dimensional Printing: A Review of the Current Literature and How to Get Started. Annals of Translational Medicine. 2016;4(23):456.
[24] Litjens G, Kooi T, Bejnordi BE, Setio AAA, Ciompi F, Ghafoorian M, van der Laak JAWM, van Ginneken B, Sánchez CI. A Survey on Deep Learning in Medical Image Analysis. Medical Image Analysis. 2017;42:60–88.
[25] Kelly CJ, Karthikesalingam A, Suleyman M, Corrado G, King D. Key Challenges for Delivering Clinical Impact With Artificial Intelligence. BMJ. 2019;364:l233.
[26] Liu X, Faes L, Kale AU, Wagner SK, Fu DJ, Bruynseels A, Mahendiran T, Moraes G, Shamdas M, Kern C, et al. A Comparison of Deep Learning Performance Against Health-Care Professionals in Detecting Diseases From Medical Imaging: A Systematic Review and Meta-Analysis. The Lancet Digital Health. 2019;1(6):e271–e297.
[27] Senders JT, Staples PC, Karhade AV, Zaki MM, Gormley WB, Broekman MLD, Smith TR. Machine Learning and Neurosurgical Outcome Prediction: A Systematic Review. World Neurosurgery. 2018;109:476–486.
[28] Collins GS, Reitsma JB, Altman DG, Moons KGM. Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD): The TRIPOD Statement. Annals of Internal Medicine. 2015;162(1):55–63.
[29] Arrieta AB, Díaz-Rodríguez N, Del Ser J, Bennetot A, Tabik S, Barbado A, García S, Gil-López S, Molina D, Benjamins R, et al. Explainable Artificial Intelligence (XAI): Concepts, Taxonomies, Opportunities and Challenges Toward Responsible AI. Information Fusion. 2020;58:82–115.
