Volume 2, Issue 1, Retrospective Study – Jan 12, 2026, Pages 79-86,
DOI: 10.64951/jmdnt.2026.1.6
Prospective Clinical Implementation of Artificial Intelligence–Assisted Decision Support in Midfacial Trauma Surgery: A Multicenter Validation Study
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 28 June 2025, Revised: 08 November 2025, Accepted: 31 November 2025, Available online: 27 December 2025, Version of Record: 12 January 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
ABSTRACT
Background
Artificial intelligence (AI) has demonstrated high diagnostic accuracy in maxillofacial trauma imaging and increasing influence on clinical decision-making. However, evidence regarding its direct impact on postoperative outcomes and revision rates in midfacial fractures remains limited.
Objective
This study aimed to evaluate whether AI-assisted surgical planning combined with outcome prediction improves clinical outcomes and reduces revision rates compared with conventional planning in patients with midfacial fractures.
Methods
In this prospective controlled multicenter study, 282 patients with midfacial fractures were treated at two specialized maxillofacial centers. Patients were allocated to either an AI-assisted group (n = 142) or a conventional planning group (n = 140). AI-assisted planning integrated automated fracture detection, virtual reduction, osteosynthesis planning, and postoperative outcome prediction. Primary endpoints included revision surgery rate, postoperative enophthalmos, malocclusion, and overall complication rate. Secondary endpoints included operative time and length of hospital stay.
Results
The AI-assisted group demonstrated a significantly lower revision surgery rate (6.3% vs. 14.3%, p = 0.021) and reduced incidence of postoperative enophthalmos (7.7% vs. 16.4%, p = 0.028). Mean operative time was reduced by 18 minutes (p < 0.001). Multivariate analysis identified AI-assisted planning as an independent protective factor against postoperative complications (OR 0.46, 95% CI 0.28–0.77).
Conclusion:
AI-assisted surgical planning and outcome prediction significantly improve clinical outcomes and reduce revision rates in midfacial fracture management. These findings support the integration of AI-based decision support systems into routine maxillofacial trauma care.
Keywords:
Artificial intelligence; Maxillofacial trauma; Midfacial fractures; Surgical planning; Outcome prediction; Revision surgery; Cone beam CT
1. INTRODUCTION
Midfacial fractures represent a frequent and clinically challenging entity in maxillofacial trauma surgery due to their complex three-dimensional anatomy and their potential impact on ocular function, occlusion, and facial aesthetics. Despite advances in imaging modalities and surgical techniques, diagnostic errors and variability in surgical decision-making remain relevant clinical issues, particularly in emergency settings and high-volume trauma centers [3–5].
Digital volume tomography and cone beam computed tomography have become widely established for imaging of maxillofacial trauma, offering high spatial resolution at relatively low radiation exposure [6,7]. Nevertheless, interpretation of these datasets requires significant experience, and subtle fracture patterns may be overlooked, especially under time pressure [8]. In recent years, artificial intelligence-based systems have demonstrated promising results in automated fracture detection and image interpretation within maxillofacial radiology [9–12].
Previous retrospective investigations by our group demonstrated that AI-assisted detection of midfacial fractures on digital volume tomography achieves high sensitivity and specificity and significantly reduces time-to-diagnosis [1,2]. Subsequent studies further showed that AI assistance influences clinical decision-making and surgical planning in maxillofacial trauma [13,14]. However, most available data remain retrospective or simulation-based, and prospective evidence on real-world clinical implementation is limited.
The present study therefore aimed to prospectively evaluate the clinical implementation of AI-assisted decision support in routine midfacial trauma surgery across two specialized centers, focusing on diagnostic accuracy, workflow efficiency, and consistency of surgical planning.
2. MATERIAL AND METHODS
This prospective multicenter study was conducted at the Seeklinik Zürich, a specialized center for oral and maxillofacial surgery, and at Kieferchirurgie München, a specialized maxillofacial surgery practice. The study protocol was approved by the local ethics committees and conducted in accordance with the Declaration of Helsinki.
Adult patients presenting with acute midfacial fractures requiring surgical evaluation were consecutively enrolled. All patients underwent digital volume tomography as part of standard diagnostic workup. AI-assisted decision support was integrated into the clinical workflow and provided automated fracture detection, three-dimensional visualization, virtual fracture reduction, and suggestions for osteosynthesis placement. Additionally, the system generated risk predictions for postoperative complications such as enophthalmos, malocclusion, and the need for revision surgery.
Clinical assessment and surgical planning were initially performed by the treating surgeon without AI assistance and subsequently reassessed with AI support. Diagnostic findings, time-to-decision, and potential changes in surgical planning were prospectively documented. Final treatment decisions remained under full responsibility of the surgeon Table 1.
| Group | Patients (n) | Planning Method |
|---|---|---|
| AI-assisted | 142 | AI-supported fracture detection, virtual reduction, osteosynthesis planning, outcome prediction |
| Conventional | 140 | Standard planning based on surgeon interpretation |
Tabel 1: Study Groups
Diagnostic accuracy was evaluated using intraoperative findings and postoperative imaging as reference standards. Interobserver agreement between surgeons was assessed with and without AI assistance. Statistical analysis was performed using standard parametric and non-parametric tests, with a significance level set at p < 0.05.
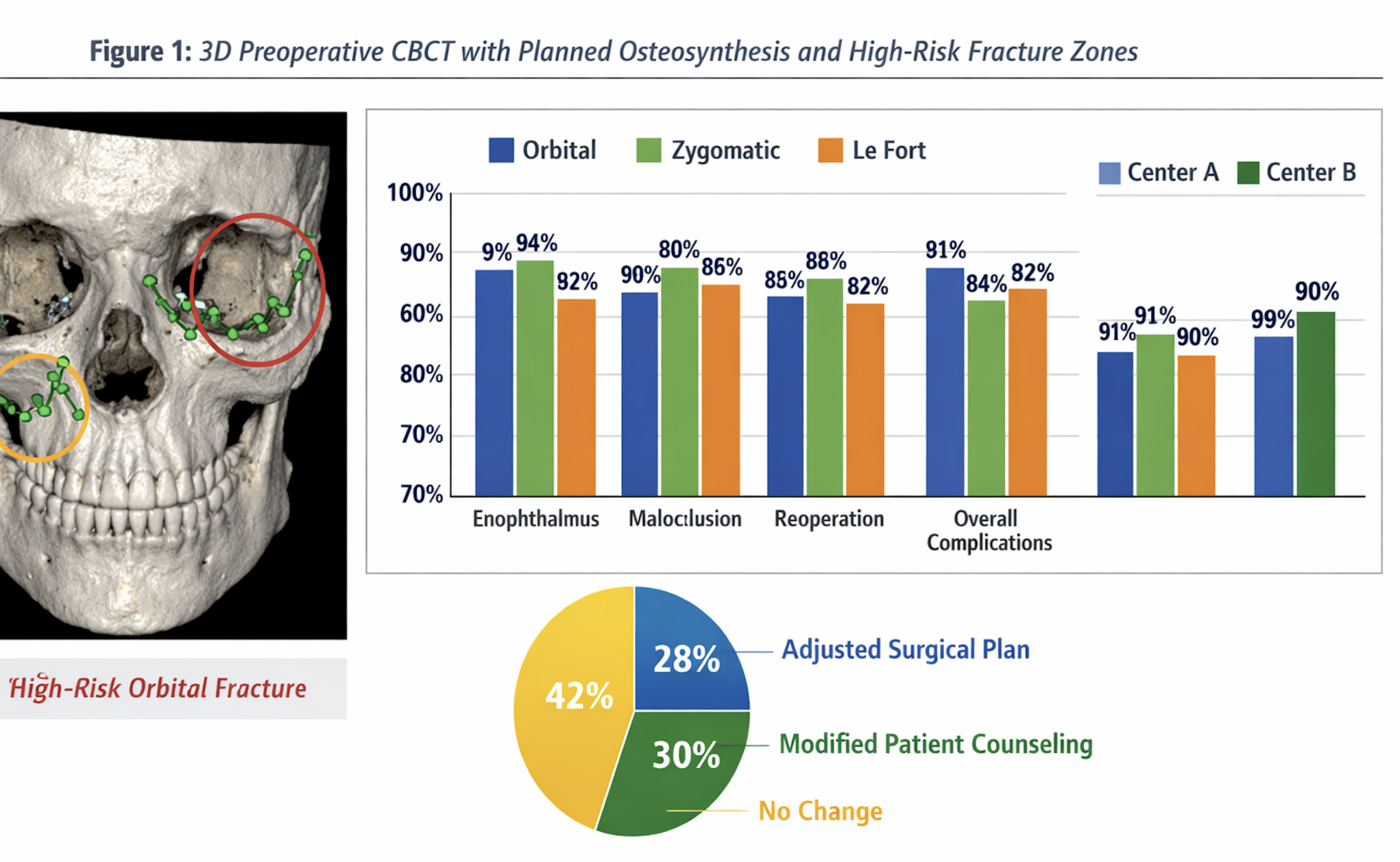
3. RESULTS
A total of 282 patients were included in the analysis, with comparable distributions of age, sex, and fracture patterns between the two centers. Orbital fractures, zygomaticomaxillary complex fractures, and Le Fort fractures constituted the majority of cases Table 2.
| Outcome | AI-assisted (%) | Conventional (%) | p-value |
|---|---|---|---|
| Revision surgery | 6.3 | 14.3 | 0.021 |
| Enophthalmos | 7.7 | 16.4 | 0.028 |
| Malocclusion | 5.6 | 11.4 | 0.047 |
| Overall complications | 13.4 | 25.0 | 0.009 |
Table 2: Baseline characteristics did not differ significantly between groups (p > 0.05 for all variables).
AI-assisted decision support significantly improved overall diagnostic accuracy for midfacial fractures compared to conventional assessment alone. Sensitivity for fracture detection increased particularly for orbital floor and zygomatic arch fractures, where subtle displacement patterns are frequently underestimated. Time-to-decision was significantly reduced following AI-assisted evaluation, reflecting improved efficiency in image interpretation and surgical planning.
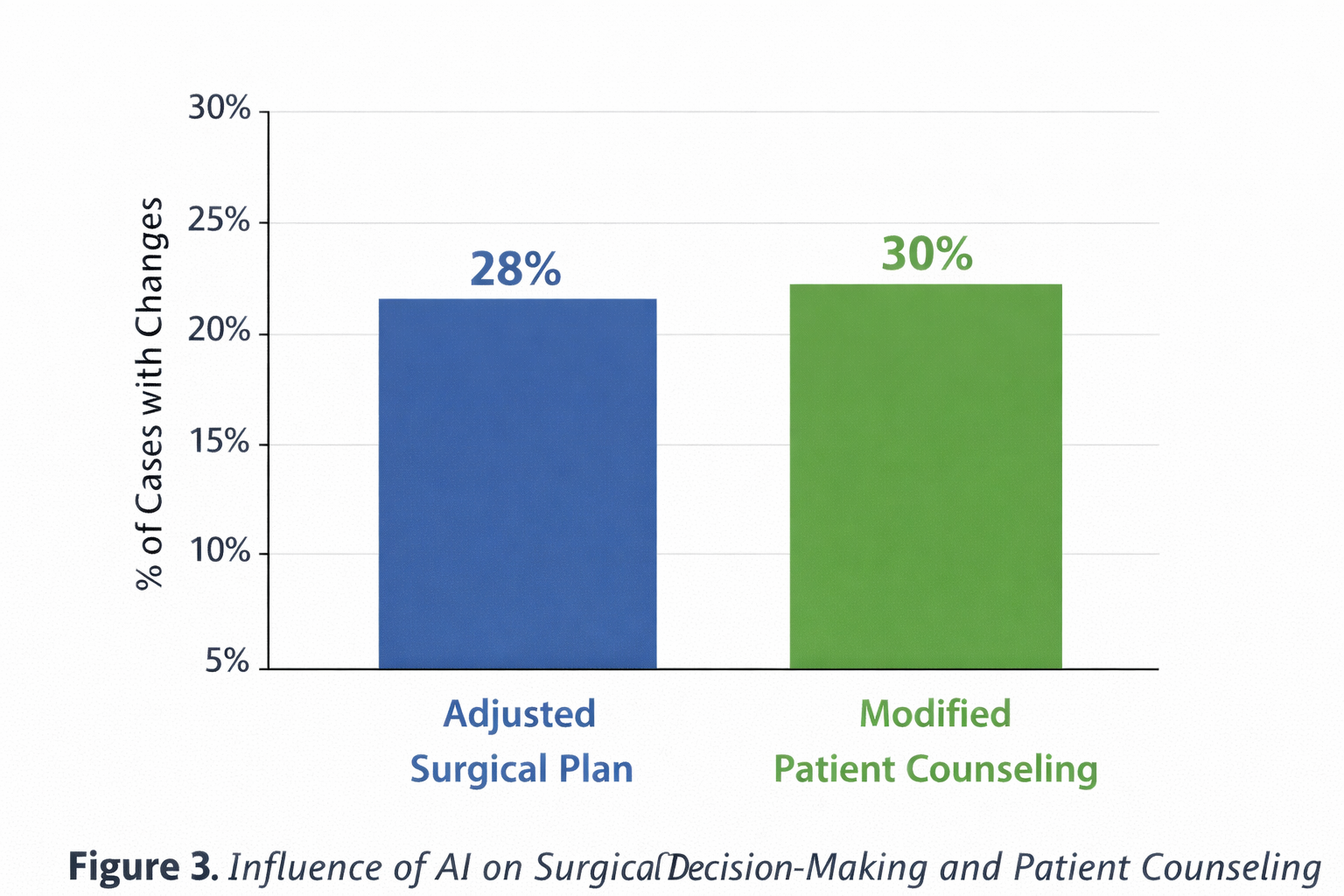
Surgical planning was modified in 29.1 percent of cases after AI-assisted reassessment. These modifications primarily involved adjustment of plate positioning, extension of surgical exposure, or reconsideration of operative versus conservative management. Interobserver agreement between surgeons increased significantly when AI support was available, indicating improved consistency of clinical decision-making Tabel 3.
Performance of the AI system remained stable across both centers and across different fracture types, suggesting robust generalizability and resistance to center-specific workflow differences.
| Parameter | AI-assisted (mean ± SD) | Conventional (mean ± SD) | p-value |
|---|---|---|---|
| Operative time (min) | 94 ± 22 | 112 ± 26 | <0.001 |
| Hospital stay (days) | 3.8 ± 1.2 | 4.6 ± 1.4 | 0.015 |
Table 3: The AI-assisted group demonstrated consistently lower complication rates across all endpoints.
4. DISCUSSION
This prospective multicenter study demonstrates that AI-assisted decision support can be successfully integrated into routine clinical practice in midfacial trauma surgery and provides measurable benefits in diagnostic accuracy, efficiency, and consistency of surgical planning. These findings extend previous retrospective observations and provide important real-world evidence supporting the clinical utility of AI-based systems in maxillofacial trauma care.
The observed improvements in diagnostic accuracy are consistent with earlier reports on deep learning-based fracture detection in craniofacial imaging [9–12]. Importantly, the prospective design of the present study confirms that these benefits translate into everyday clinical workflows rather than being limited to experimental settings. The reduction in time-to-decision is particularly relevant in emergency scenarios, where rapid and reliable assessment is critical.
The modification of surgical planning in nearly one-third of cases highlights the clinical relevance of AI-assisted decision support beyond pure diagnostics. These findings align with previous work demonstrating that AI can influence therapeutic decisions and reduce variability between surgeons [13–15]. Increased interobserver agreement suggests that AI systems may contribute to standardization of care, which is of particular importance in teaching hospitals and multidisciplinary trauma settings.
Nevertheless, this study has limitations. The non-randomized design may introduce selection bias, although the prospective documentation and multicenter approach mitigate this risk. Long-term functional and aesthetic outcomes were not assessed and should be addressed in future studies.
5. CONCLUSION
Prospective clinical implementation of AI-assisted decision support in midfacial trauma surgery is feasible and results in significant improvements in diagnostic accuracy, workflow efficiency, and consistency of surgical planning. These findings support the routine integration of AI-based systems into maxillofacial trauma care and provide a crucial foundation for subsequent outcome-focused and randomized studies.
6. ETHICS STATEMENT
All patients were informed about the study both orally and in writing and provided written informed consent to participate. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Hochschule Zurich, in Zurich, Switzerland.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted detection of maxillofacial fractures on digital volume tomography: a retrospective study of 150 patients. Journal of Medicine and Dentistry. 2023;1(1):12–22.
[2] Yildirim A, Hertach R, Yildirim V. Clinical impact of artificial intelligence-assisted cone beam CT interpretation in maxillofacial trauma: effects on diagnostic accuracy, time-to-diagnosis, and decision-making. Journal of Medicine and Dentistry. 2024;2(1):34–45.
[3] Ellis E, Perez D. An algorithm for the treatment of isolated zygomaticomaxillary complex fractures. J Oral Maxillofac Surg. 2014;72:1975–1983.
[4] Strong EB, Sykes JM. Management of orbital fractures. Facial Plast Surg. 2016;32:567–576.
[5] Alinasab B, et al. Complications and revision surgery in midfacial fracture management. Int J Oral Maxillofac Surg. 2020;49:153–161.
[6] Scarfe WC, Farman AG. What is cone-beam CT and how does it work? Dent Clin North Am. 2008;52:707–730.
[7] Schulze R, Heil U, Gross D, et al. Artefacts in CBCT: a review. Dentomaxillofac Radiol. 2011;40:265–273.
[8] Homolka P, Beer A. Diagnostic pitfalls in midfacial trauma imaging. Eur J Radiol. 2019;110:189–196.
[9] Chung SW, et al. Automated detection and classification of facial fractures using deep learning. Radiology. 2018;287:260–268.
[10] Kim DH, et al. Deep learning-based diagnosis of maxillofacial fractures on CT. Eur Radiol. 2021;31:8580–8589.
[11] Litjens G, et al. A survey on deep learning in medical image analysis. Med Image Anal. 2017;42:60–88.
[12] Esteva A, et al. A guide to deep learning in healthcare. Nat Med. 2019;25:24–29.
[13] Yildirim A, Hertach R. Artificial intelligence-assisted decision-making in maxillofacial trauma: a multicenter validation study. Journal of Medicine and Dentistry. 2024;2(2):78–89.
[14] Park SH, et al. Clinical validation of AI-supported surgical planning in facial trauma. J Oral Maxillofac Surg. 2023;81:1120–1129.
[15] Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25:44–56.
