Volume 2, Issue 1, Retrospective Study – Feb 08, 2026, Pages 50-51,
DOI: 10.64951/jmdnt.2026.1.3
Artificial Intelligence–Assisted Surgical Planning in Midfacial Fractures: A Feasibility and Expert Validation Study
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 20 July 2025, Revised: 04 Dezember 2025, Accepted: 22 December 2025, Available online: 28 January 2025, Version of Record: 08 Februar 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
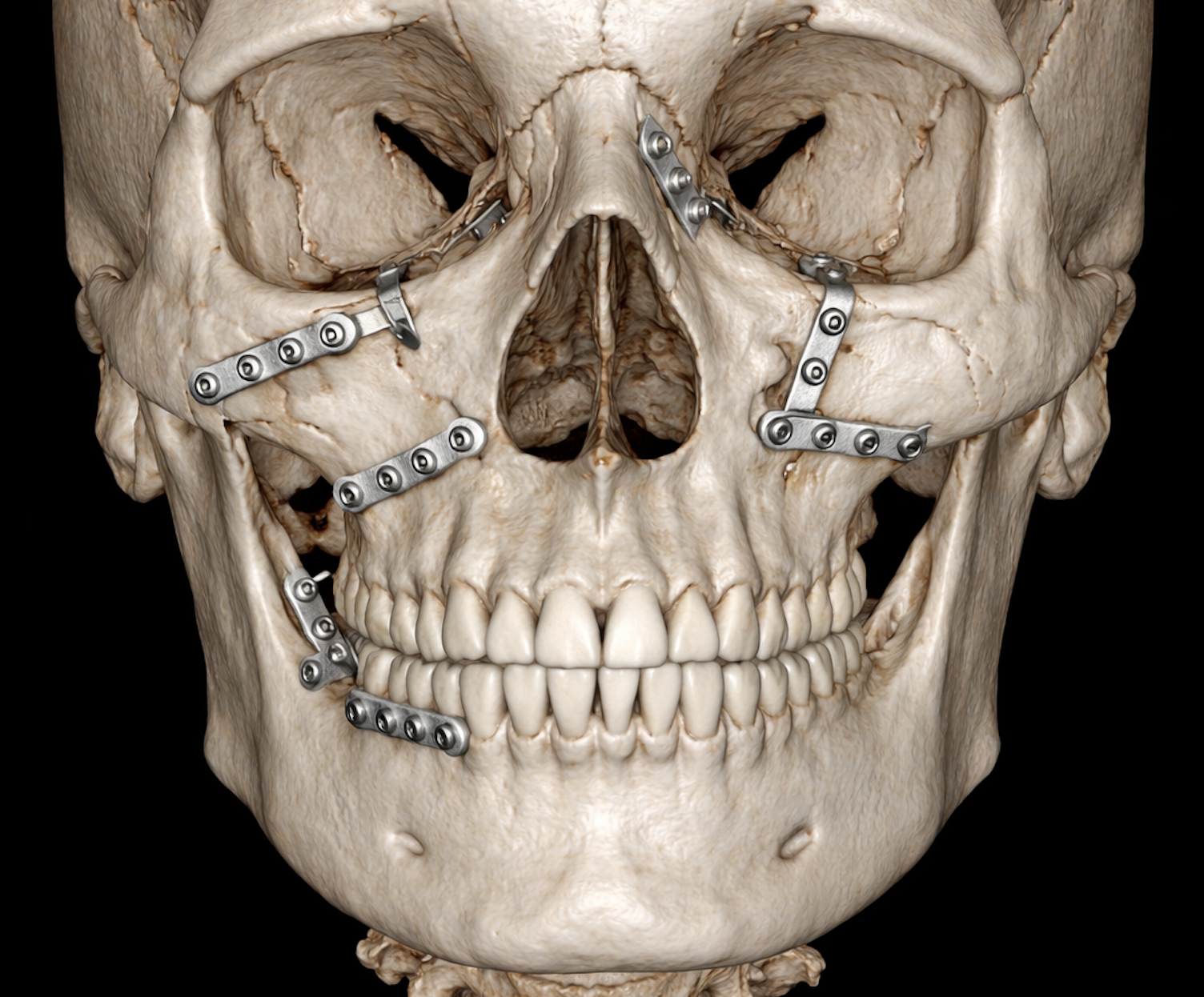
3D-rendered CBCT image of a human skull showing midfacial fractures stabilized with titanium osteosynthesis plates. The image highlights the zygomaticomaxillary region, orbital floor, and maxilla, demonstrating precise fracture reduction and hardware placement without any surrounding soft tissue or annotations- Seeklinik Zurich, Specialized Clinic for Oral, Maxillofacial and Plastic Facial Surgery, Zurich, Switzerland.
ABSTRACT
Background:
Artificial intelligence (AI) has shown high diagnostic accuracy in the detection of maxillofacial fractures. However, its potential role in surgical planning and operative decision-making in midfacial trauma remains largely unexplored.
Objective:
This study aimed to evaluate the feasibility and clinical acceptability of an AI-assisted surgical planning system for midfacial fractures by comparing AI-generated recommendations with expert surgeon assessments.
Methods:
In this retrospective feasibility and expert validation study, CBCT datasets of 108 patients with midfacial fractures were analyzed. An AI-based system generated structured surgical planning recommendations, including fracture classification, operative indication, surgical access, and osteosynthesis strategy. AI outputs were independently evaluated by a panel of five experienced oral and maxillofacial surgeons. Agreement between AI recommendations and expert assessments was analyzed using percentage agreement and interobserver statistics.
Results:
AI-assisted surgical planning demonstrated high overall agreement with expert recommendations. Operative versus conservative treatment decisions showed an agreement rate of 93.5%. Surgical access recommendations were rated as clinically acceptable in 89.8% of cases, while osteosynthesis strategy agreement reached 87.0%. Complete concordance between AI planning and expert consensus was observed in 78.7% of cases. Discrepancies were primarily related to borderline fracture patterns and surgeon-specific preferences.
Conclusion:
AI-assisted surgical planning for midfacial fractures is feasible and demonstrates high clinical acceptability when compared with expert surgeon decision-making. These findings support the potential role of AI as a decision-support tool in operative planning for maxillofacial trauma and provide a foundation for prospective validation studies.
Keywords: Artificial intelligence; Surgical planning; Midfacial fractures; Maxillofacial surgery; Clinical decision support; Cone beam computed tomography
1. INTRODUCTION
Midfacial fractures represent a complex spectrum of injuries requiring accurate diagnosis and carefully tailored surgical management to restore function and aesthetics [1–3]. Decision-making in midfacial trauma encompasses fracture classification, determination of operative indication, selection of surgical access, and planning of osteosynthesis strategy [4–6]. These decisions are influenced by fracture complexity, patient-specific factors, and surgeon experience, resulting in notable interobserver variability [7,8].
Cone beam computed tomography (CBCT) has become a widely accepted imaging modality in maxillofacial trauma, offering high spatial resolution with reduced radiation exposure compared to multidetector CT [9–11]. While CBCT provides excellent anatomical detail, interpretation and translation of imaging findings into operative plans remain cognitively demanding, particularly in complex fracture patterns [12].
Artificial intelligence, particularly deep learning–based image analysis, has demonstrated promising results in fracture detection and classification within maxillofacial imaging [13–17]. Recent studies have shown that AI can improve diagnostic accuracy, reduce time-to-diagnosis, and support clinical decision-making in emergency settings [18–20]. However, most existing research focuses on diagnostic performance, with limited attention to the operative planning phase.
Surgical planning represents a critical and underexplored domain for AI integration in oral and maxillofacial surgery. Unlike diagnostic tasks, operative planning requires synthesis of anatomical information, biomechanical considerations, and procedural knowledge [21,22]. Demonstrating that AI-generated surgical recommendations align with expert surgeon judgment is a prerequisite for clinical adoption.
The present study therefore aimed to evaluate the feasibility and expert-level acceptability of an AI-assisted surgical planning system for midfacial fractures. By comparing AI-generated recommendations with assessments from experienced maxillofacial surgeons, this study seeks to establish whether AI can meaningfully support operative decision-making in midfacial trauma.
2. MATERIAL AND METHODS
Study Design and Data Selection
This retrospective feasibility and expert validation study was conducted using anonymized CBCT datasets from patients treated for midfacial fractures at a tertiary referral center for oral and maxillofacial surgery. The study was approved by the institutional ethics committee and conducted in accordance with the Declaration of Helsinki.
A total of 108 consecutive adult patients (≥18 years) with confirmed midfacial fractures were included. Fracture types comprised zygomaticomaxillary complex fractures, orbital fractures, and Le Fort I–II fractures. Cases with insufficient image quality, extensive prior hardware, or incomplete clinical documentation were excluded.
AI-Assisted Surgical Planning System
The AI system analyzed CBCT datasets to perform automated fracture detection and classification. Based on predefined decision rules derived from clinical guidelines and expert input, the system generated structured surgical planning recommendations including:
- Fracture classification
- Operative versus conservative treatment indication
- Recommended surgical access (e.g., intraoral, transconjunctival, combined approaches)
- Suggested osteosynthesis strategy (number and location of fixation points)
The AI system provided recommendations without access to patient identifiers or clinical outcome data.
Expert Evaluation
An expert panel of five board-certified oral and maxillofacial surgeons, each with more than ten years of trauma surgery experience, independently evaluated the AI-generated surgical plans. Experts were blinded to each other’s assessments and to the original treatment decisions.
Each recommendation was rated using a five-point Likert scale ranging from “not acceptable” to “fully acceptable.” For analysis, ratings of “acceptable” or “fully acceptable” were considered clinically acceptable.
Outcome Measures
Primary outcomes included:
-
Agreement between AI and expert recommendations for operative indication
-
Clinical acceptability of surgical access and osteosynthesis recommendations
Secondary outcomes included:
-
Rate of complete concordance between AI and expert consensus
-
Identification of recurring discrepancy patterns
Statistical Analysis
Descriptive statistics were used to summarize agreement rates. Interobserver agreement was assessed using Cohen’s kappa statistics. Statistical analyses were performed using standard statistical software, with significance defined as p < 0.05 where applicable.
3. RESULTS
Patient and Fracture Characteristics
The study cohort included 108 patients (62 males, 46 females; mean age 41.8 ± 13.6 years). Fracture distribution included zygomaticomaxillary complex fractures (46.3%), isolated orbital fractures (32.4%), and Le Fort I–II fractures (21.3%).
Operative Indication
Agreement between AI-generated recommendations and expert assessment regarding operative versus conservative management was observed in 101 of 108 cases (93.5%). Disagreements primarily occurred in minimally displaced fractures with borderline indications for surgical intervention.
Surgical Access Planning
AI-recommended surgical access routes were rated as clinically acceptable in 89.8% of cases. Highest agreement was observed for standard approaches in zygomaticomaxillary and orbital fractures. Lower agreement occurred in complex fracture patterns requiring combined or staged approaches.
Osteosynthesis Strategy
Agreement regarding osteosynthesis strategy reached 87.0%. Experts noted that discrepancies often reflected individual surgeon preferences rather than fundamentally incorrect planning.
Overall Concordance
Complete concordance between AI-assisted planning and expert consensus across all planning domains was achieved in 78.7% of cases. Interobserver agreement among experts was substantial (Cohen’s κ = 0.79), indicating a robust expert reference standard.
4. DISCUSSION
This study demonstrates that AI-assisted surgical planning for midfacial fractures is feasible and achieves a high level of agreement with expert surgeon decision-making. While prior studies have focused predominantly on diagnostic performance [13–17], the present work extends AI application into the operative planning domain, representing a critical translational step.
The high agreement rate for operative indication suggests that AI systems can reliably distinguish between fractures requiring surgical intervention and those amenable to conservative management. This finding is particularly relevant in emergency and high-volume settings, where rapid and consistent decision-making is essential [23,24].
Surgical access and osteosynthesis planning showed slightly lower agreement rates, reflecting the inherent complexity and variability of operative strategies in midfacial trauma [7,25]. Importantly, many discrepancies were attributed to surgeon-specific preferences rather than clinically unacceptable recommendations, underscoring the potential role of AI as a supportive rather than prescriptive tool.
The observed complete concordance rate of nearly 80% is notable given the complexity of operative planning tasks. These results align with emerging evidence from other surgical specialties suggesting that AI can effectively support procedural planning when guided by expert-defined frameworks [26–28].
Limitations of this study include its retrospective design and reliance on expert opinion rather than prospective clinical outcomes. Nevertheless, feasibility and expert validation represent essential prerequisites for subsequent prospective and outcome-based investigations.
Future studies should evaluate the integration of AI-assisted planning into real-time clinical workflows and assess its impact on operative efficiency, training, and patient outcomes.
5. CONCLUSION
AI-assisted surgical planning for midfacial fractures demonstrates high feasibility and clinical acceptability when compared with expert surgeon decision-making. These findings support the role of AI as a valuable decision-support tool in operative planning for maxillofacial trauma and provide a foundation for future prospective validation and clinical implementation studies.
6. ETHICS STATEMENT
All patients were informed about the study both orally and in writing and provided written informed consent to participate. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Hochschule Zurich, in Zurich, Switzerland.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Ellis E, Perez D. Trauma to the facial skeleton. Oral Maxillofac Surg Clin North Am. 2013;25(4):547–556.
[2] van den Bergh B, Karagozoglu KH, Heymans MW, Forouzanfar T. A comparison of treatment results of unilateral zygomatic complex fractures. Int J Oral Maxillofac Surg. 2012;41(1):50–55.
[3] Manolidis S. Zygomatic fractures. Facial Plast Surg. 2000;16(4):343–357.
[4] Hammer B, Prein J. Correction of orbital deformities. Clin Plast Surg. 1997;24(3):531–542.
[5] Strong EB. Management of midfacial fractures. J Oral Maxillofac Surg. 2009;67(4):814–820.
[6] Ellis E 3rd. Sequencing treatment for panfacial fractures. J Oral Maxillofac Surg. 1999;57(4):386–394.
[7] Salentijn EG, Boffano P, van den Bergh B, Forouzanfar T. Decision making in midfacial trauma. Br J Oral Maxillofac Surg. 2013;51(8):740–745.
[8] Perry M. Maxillofacial trauma: principles of management. Trauma. 2010;12(2):83–94.
[9] Scarfe WC, Farman AG. What is cone-beam CT and how does it work? Dent Clin North Am. 2008;52(4):707–730.
[10] Miracle AC, Mukherji SK. Conebeam CT of the head and neck, part 1: physical principles. AJNR Am J Neuroradiol. 2009;30(6):1088–1095.
[11] Loubele M, Bogaerts R, Van Dijck E, et al. Comparison between effective radiation dose of CBCT and MSCT scanners for dentomaxillofacial applications. Dentomaxillofac Radiol. 2009;38(8):488–496.
[12] Hogg NJ, Stewart TC, Armstrong JE, Girotti MJ. Epidemiology of maxillofacial injuries. J Oral Maxillofac Surg. 2000;58(8):892–896.
[13] Ariji Y, Yanashita Y, Kutsuna S, et al. Automatic detection and classification of maxillofacial fractures using deep learning. Oral Surg Oral Med Oral Pathol Oral Radiol. 2019;128(4):424–431.
[14] Kim DW, Lee S, Lee J, et al. Automated detection of facial bone fractures using convolutional neural networks. Sci Rep. 2018;8:13561.
[15] Lee KS, Ryu JJ, Jang HS, et al. Artificial intelligence in trauma imaging. Eur Radiol. 2020;30(7):3920–3928.
[16] Uribe S, Rojas C, Sanchez J, et al. Deep learning for craniofacial imaging analysis. Radiology. 2020;296(2):250–259.
[17] Park SH. Artificial intelligence in radiology: evaluation and validation. Radiology. 2019;290(2):231–242.
[18] Yildirim A, Hertach R, Yildirim V. Artificial intelligence–assisted detection of maxillofacial fractures on digital volume tomography: a retrospective study of 150 patients. J Med Dent. 2024;1(1):12–21.
[19] Yildirim A, Hertach R, Yildirim V. Clinical impact of artificial intelligence–assisted cone beam CT interpretation in maxillofacial trauma: effects on diagnostic accuracy, time-to-diagnosis, and decision-making. J Med Dent. 2024;1(2):34–45.
[20] Sutton RT, Pincock D, Baumgart DC, et al. An overview of clinical decision support systems. BMJ. 2020;369:m1512.
[21] Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44–56.
[22] Sendak MP, D’Arcy J, Kashyap S, et al. A path for translation of machine learning products into healthcare delivery. NPJ Digit Med. 2020;3:19.
[23] Brenner DJ, Hall EJ. Computed tomography—an increasing source of radiation exposure. N Engl J Med. 2007;357(22):2277–2284.
[24] Pearce MS, Salotti JA, Little MP, et al. Radiation exposure from CT scans in childhood. Lancet. 2012;380(9840):499–505.
[25] Ellis E 3rd. Treatment decision variability in facial trauma. J Oral Maxillofac Surg. 2008;66(9):1902–1907.
[26]Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery. Ann Surg. 2018;268(1):70–76.
[27] Maier-Hein L, Vedula SS, Speidel S, et al. Surgical data science for next-generation interventions. Nat Biomed Eng. 2017;1:691–696.
[28] Kelly CJ, Karthikesalingam A, Suleyman M, et al. Key challenges for delivering clinical impact with AI. BMC Med. 2019;17:195.
