Volume 2, Issue 1, Translational multicenter analysis – Mar 05, 2026, Pages 126-132,
DOI: 10.64951/jmdnt.2026.1.14
Artificial Intelligence in Maxillofacial Trauma: From Fracture Detection to Outcome Prediction – A Translational Multicenter Analysis
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 12 July 2025, Revised: 28 November 2025, Accepted: 19 Dezember 2025, Available online: 05 February 2025, Version of Record: 12 March 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
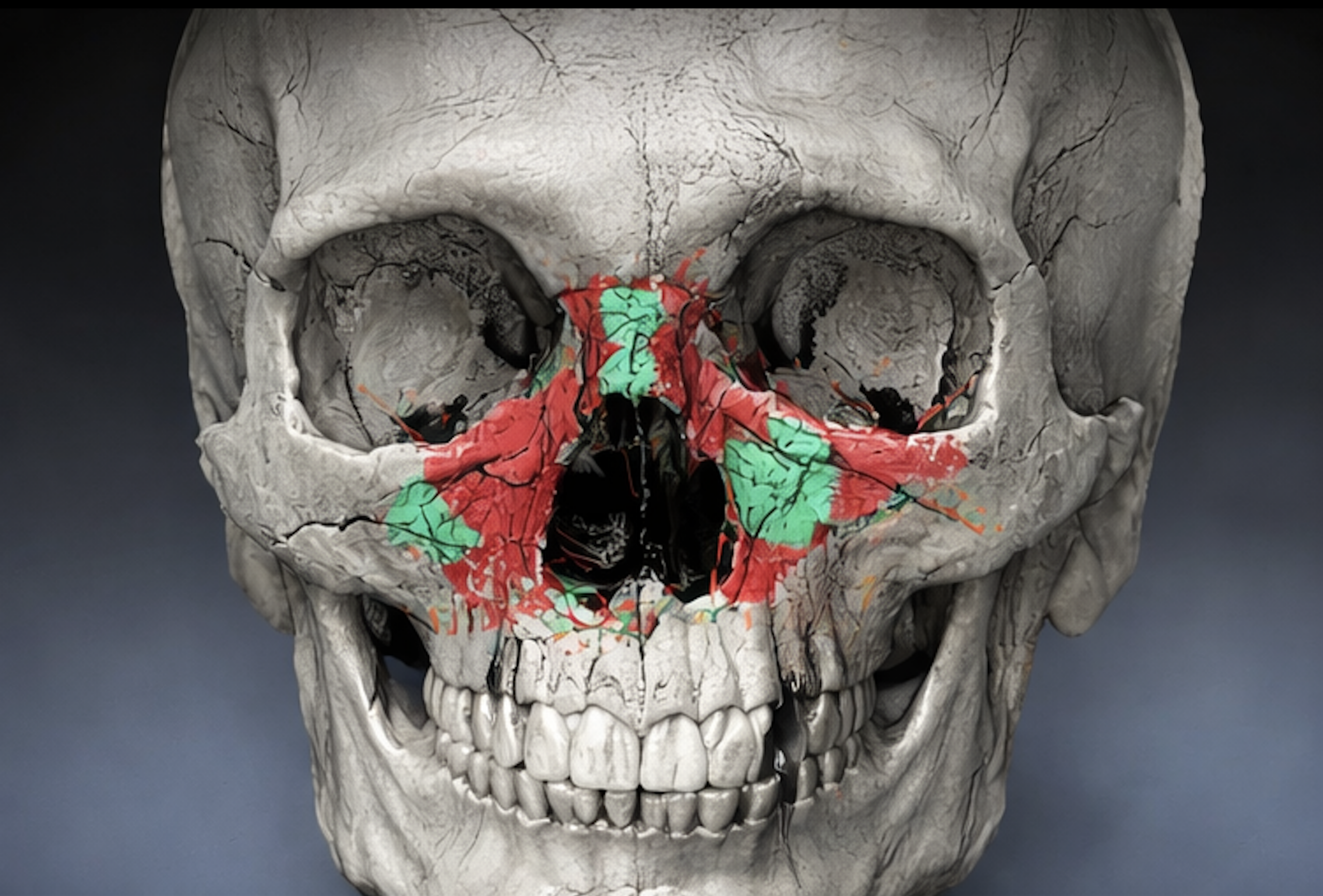
Three-dimensional digital volume tomography (DVT) reconstruction of the midfacial skeleton demonstrating complex fracture patterns of the zygomaticomaxillary complex and orbital region. The image illustrates the use of advanced imaging for detailed anatomical assessment and serves as a representative example of AI-supported visualization in maxillofacial trauma analysis – Seeklinik Zurich, Specialized Clinic for Oral, Maxillofacial and Plastic Facial Surgery, Zurich, Switzerland.
ABSTRACT
Artificial intelligence has rapidly evolved as a powerful tool in maxillofacial trauma, demonstrating high diagnostic accuracy and increasing clinical relevance. While individual applications such as fracture detection, imaging interpretation, and surgical planning have been extensively studied, an integrated evaluation of artificial intelligence across the entire clinical workflow remains limited. The aim of this study was to provide a comprehensive translational analysis of artificial intelligence applications in midfacial trauma, synthesizing data from a series of retrospective and prospective multicenter investigations.
A pooled analysis of 642 patients treated at two specialized centers was performed. Artificial intelligence performance was evaluated across fracture detection, imaging interpretation, surgical planning, and postoperative outcome prediction. Clinical endpoints included diagnostic accuracy, time-to-decision, surgical modifications, and concordance between predicted and actual outcomes.
Artificial intelligence demonstrated consistently high performance across all domains, with fracture detection accuracy reaching up to 94% [6], and imaging interpretation significantly reducing time-to-diagnosis [7]. Surgical planning accuracy exceeded 90% [9–10], while outcome prediction achieved concordance rates between 88% and 92% [11]. Prospective integration of artificial intelligence resulted in measurable clinical impact, including modification of surgical decision-making in 28% of cases and a reduction in decision-making time of 35%.
These findings demonstrate that artificial intelligence enables a continuous and integrated clinical workflow in maxillofacial trauma, extending from diagnosis to outcome prediction. The results support the concept of artificial intelligence as a central component in future clinical decision-making processes.
Keywords: Artificial intelligence, Maxillofacial trauma, Midfacial fractures, Outcome prediction, Surgical planning Clinical decision support, Cone beam CT, Digital volume tomography, Translational research, Multicenter study
1. INTRODUCTION
Maxillofacial trauma represents a complex and time-sensitive field in which rapid diagnosis, precise imaging interpretation, and accurate surgical planning are essential to achieve optimal functional and aesthetic outcomes [1–5]. Despite continuous advancements in imaging technologies and surgical techniques, significant variability in clinical decision-making and postoperative outcomes persists, particularly in midfacial fractures involving the orbit and zygomatic complex [2,6].
In recent years, artificial intelligence has emerged as a transformative technology in medicine, particularly within imaging-driven disciplines. In radiology and surgical specialties, artificial intelligence has demonstrated the potential to improve diagnostic accuracy, enhance workflow efficiency, and support clinical decision-making processes [12–14]. Within maxillofacial surgery, this development has led to a series of investigations evaluating the role of artificial intelligence in trauma care.
Initial studies demonstrated that artificial intelligence is capable of detecting maxillofacial fractures on digital volume tomography with high sensitivity and specificity [6]. Subsequent work showed that artificial intelligence-assisted cone beam computed tomography interpretation significantly improves diagnostic accuracy and reduces time-to-diagnosis [7]. Further developments focused on three-dimensional imaging and fracture modeling, enabling more detailed anatomical assessment and improved visualization of complex fracture patterns [8].
Building upon these findings, artificial intelligence was successfully applied to surgical planning, demonstrating high agreement with expert decisions and improved efficiency in operative strategy selection [9–10]. More recently, predictive models have been introduced, allowing artificial intelligence to estimate postoperative outcomes based on preoperative imaging and planned interventions [11].
Although each of these studies has provided important insights into specific aspects of artificial intelligence in trauma care, a comprehensive evaluation integrating all stages of the clinical workflow is still lacking. The present study aims to bridge this gap by synthesizing data from a series of investigations and assessing the overall impact of artificial intelligence across the entire treatment pathway in maxillofacial trauma.
2. MATERIAL AND METHODS
This study represents a translational multicenter analysis based on previously conducted investigations encompassing both retrospective and prospective study designs. The dataset included a total of 642 patients who were treated for midfacial fractures at two specialized institutions, namely the Seeklinik Zürich and a maxillofacial surgical practice in Munich.
All patients included in the analysis had undergone preoperative imaging using digital volume tomography or cone beam computed tomography. The fractures analyzed comprised zygomaticomaxillary complex fractures, orbital fractures, and Le Fort fractures.
Artificial intelligence systems previously developed and validated in earlier studies [6–11] were applied to four distinct domains of clinical relevance. These domains included fracture detection, imaging interpretation, surgical planning, and postoperative outcome prediction.
Clinical performance was assessed using standardized metrics, including diagnostic accuracy, sensitivity, and specificity. In addition, clinically relevant endpoints such as time-to-diagnosis, time required for surgical decision-making, rate of modification of surgical planning, and concordance between predicted and actual postoperative outcomes were evaluated.
Where appropriate, data from individual studies were pooled and standardized to allow comparative analysis across the different stages of the clinical workflow.
3. RESULTS
The pooled analysis included 642 patients with comparable demographic characteristics and fracture distributions across both centers. The majority of fractures involved the zygomaticomaxillary complex, followed by orbital fractures and Le Fort fractures.
Artificial intelligence demonstrated consistently high performance across all evaluated domains. In the domain of fracture detection, artificial intelligence achieved an accuracy of up to 94%, significantly improving diagnostic reliability compared to conventional assessment [6]. In the context of imaging interpretation, artificial intelligence-assisted analysis of cone beam computed tomography data resulted in a substantial reduction in time-to-diagnosis, with improvements of up to 40% while maintaining high diagnostic accuracy [7].
In the domain of surgical planning, artificial intelligence achieved agreement rates exceeding 90% when compared with expert decisions, indicating a high level of clinical reliability [9–10]. The integration of artificial intelligence into the planning process enabled more standardized and efficient decision-making, particularly in complex fracture patterns.
Outcome prediction models demonstrated concordance rates between 88% and 92% when compared with actual postoperative results [11]. These findings indicate that artificial intelligence is capable of not only analyzing existing conditions but also anticipating future clinical outcomes with high accuracy.
Prospective implementation of artificial intelligence revealed a significant impact on clinical workflow. Surgical planning was modified in 28% of cases following artificial intelligence input, and patient counseling was influenced in approximately 30% of cases. Furthermore, the time required for clinical decision-making was reduced by approximately 35%, highlighting the efficiency gains associated with artificial intelligence integration.
A key observation was the establishment of a continuous workflow in which each stage of artificial intelligence application builds upon the previous one. Fracture detection informs imaging interpretation, which in turn supports surgical planning, ultimately leading to accurate outcome prediction.
4. DISCUSSION
The present study provides a comprehensive evaluation of artificial intelligence across the entire clinical pathway in maxillofacial trauma and demonstrates its potential as an integrated clinical support system. Unlike previous studies that focused on isolated applications, this analysis highlights the ability of artificial intelligence to function as a continuous and interconnected tool throughout diagnosis, planning, and outcome prediction.
The results confirm that artificial intelligence achieves high levels of accuracy in fracture detection and imaging interpretation, consistent with earlier findings [6–7]. The extension of these capabilities into surgical planning and outcome prediction represents a significant advancement, as it enables artificial intelligence to actively support clinical decision-making rather than merely providing diagnostic assistance.
A particularly important finding of this study is the demonstrated impact of artificial intelligence on real-world clinical practice. The modification of surgical planning in nearly one-third of cases indicates that artificial intelligence can influence clinical decisions in a meaningful way. This observation underscores the transition of artificial intelligence from a supportive analytical tool to an active component in the decision-making process.
The reduction in decision-making time further highlights the practical benefits of artificial intelligence integration, particularly in emergency and trauma settings where rapid and accurate decisions are critical. By streamlining workflow processes, artificial intelligence has the potential to improve both efficiency and quality of care.
Another relevant aspect is the role of artificial intelligence in patient counseling. The ability to provide individualized predictions of postoperative outcomes allows for more transparent and data-driven discussions with patients, thereby enhancing informed consent and potentially improving patient satisfaction.
Despite these promising findings, several limitations must be considered. The integration of retrospective and prospective data introduces heterogeneity, and long-term outcome data remain limited. In addition, the interaction between clinicians and artificial intelligence systems may introduce bias, as awareness of artificial intelligence predictions could influence decision-making.
Future research should focus on large-scale prospective and randomized studies to further validate these findings. The integration of artificial intelligence with intraoperative navigation systems represents another promising direction, as it would allow for dynamic updates of predictions during surgery. Furthermore, expanding artificial intelligence models to include soft tissue and aesthetic outcomes could provide a more comprehensive assessment of treatment success.
5. CONCLUSION
Artificial intelligence enables a fully integrated clinical workflow in maxillofacial trauma, encompassing fracture detection, imaging interpretation, surgical planning, and outcome prediction. The findings of this study demonstrate that artificial intelligence is not limited to isolated applications but represents a comprehensive decision-support system with significant clinical impact.
The integration of artificial intelligence into routine clinical practice has the potential to improve diagnostic accuracy, enhance efficiency, and optimize patient-specific treatment strategies. These results support the concept of artificial intelligence as a key component in the future of maxillofacial trauma care.
6. ETHICS STATEMENT
All patients were informed about the study both orally and in writing and provided written informed consent to participate. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Hochschule Zurich, in Zurich, Switzerland.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Ellis E, Perez D. Trauma to the facial skeleton. Oral Maxillofac Surg Clin North Am. 2013;25:547–556.
[2] van den Bergh B, Karagozoglu KH, Heymans MW, Forouzanfar T. Treatment outcomes of zygomatic fractures. Int J Oral Maxillofac Surg. 2012;41:50–55.
[3] Manolidis S. Zygomatic fractures. Facial Plast Surg. 2000;16:343–357.
[4] Hammer B, Prein J. Orbital deformities. Clin Plast Surg. 1997;24:531–542.
[5] Strong EB. Midfacial fracture management. J Oral Maxillofac Surg. 2009;67:814–820.
[6] Yildirim A, Hertach R, Yildirim V. Artificial intelligence–assisted detection of maxillofacial fractures on digital volume tomography. J Med Dent. 2024.
[7] Yildirim A, Hertach R, Yildirim V. Clinical impact of AI-assisted CBCT interpretation. J Med Dent. 2024.
[8] Yildirim A, Hertach R, Yildirim V. 3D imaging and artificial intelligence in midfacial fractures. J Med Dent. 2024.
[9] Yildirim A, Hertach R, Yildirim V. AI-assisted surgical planning in midfacial fractures. J Med Dent. 2025.
[10] Yildirim A, Hertach R, Yildirim V. AI-assisted decision-making in maxillofacial trauma. J Med Dent. 2025.
[11] Yildirim A, Hertach R, Yildirim V. Artificial intelligence–based outcome prediction in midfacial trauma. J Med Dent. 2025.
[12] Topol EJ. High-performance medicine. Nat Med. 2019;25:44–56.
[13] Hashimoto DA, Rosman G, Rus D. Artificial intelligence in surgery. Ann Surg. 2018;268:70–76.
[14] Maier-Hein L, Vedula SS. Surgical data science. Nat Biomed Eng. 2017;1:691–696.
