Volume 3, Issue 2, Prospective Multicenter Analysis – Apr 13, 2026, Pages 54-64,
DOI: 10.64951/jmdnt.2026.2.19
Cost-Effectiveness and Health Economic Impact of AI-Guided 3D-Printed Surgical Workflows in Maxillofacial Trauma Surgery: A Prospective Multicenter Analysis
Ayhan Yildirim¹, René Hertach², Vedat Yildirim¹
¹ Hochschule Zurich, Department of Medicine, Albisstrasse 80, 8038 Zurich, Switzerland
² Hochschule Zurich, Department of Dentistry, Albisstrasse 80, 8038 Zurich, Switzerland
Received: 12 January 2026, Revised: 20 February 2026, Accepted: 10 March 2026, Available online: 13 April 2026, Version of Record: 13 April 2026
© 2026 Journal of Medicine and Dentistry (JMDNT)
This article is published under the Creative Commons Attribution 4.0 International (CC BY 4.0) License.
You are free to share and adapt the material for any purpose, even commercially, as long as proper credit is given to the original author(s) and source.
Full license details
ABSTRACT
Background:
Artificial intelligence (AI) and additive manufacturing have been shown to improve diagnostic accuracy, surgical planning, and intraoperative precision in maxillofacial trauma surgery [1–10]. While previous studies demonstrated improved surgical accuracy, reduced operative time, and favorable long-term outcomes, the economic impact of AI-guided workflows remains insufficiently investigated.
Objective:
This study aimed to evaluate the cost-effectiveness and health economic impact of AI-guided three-dimensional (3D) printed surgical workflows compared with conventional surgical approaches in maxillofacial trauma surgery.
Methods:
A prospective multicenter analysis was conducted across two maxillofacial surgery centers. Patients undergoing surgical treatment for maxillofacial fractures were assigned to either an AI-guided 3D-printed surgical guide workflow or a conventional surgical workflow. Direct and indirect costs, operative time, length of hospital stay, complication rates, and revision surgery rates were assessed. Cost-effectiveness was evaluated using cost per patient, cost per avoided complication, and cost per avoided revision surgery.
Results:
A total of 128 patients were included (AI-guided: n=64; conventional: n=64). Mean operative time was significantly reduced in the AI-guided group (−21.4%, p<0.001). Despite additional costs for guide fabrication, total treatment costs per patient were reduced by 14.8% due to shorter operative time, reduced complication rates, and lower revision rates. Cost per avoided revision surgery was substantially lower in the AI-guided group. Length of hospital stay was reduced by 1.2 days on average.
Conclusion:
AI-guided 3D-printed surgical workflows are not only clinically effective but also economically advantageous in maxillofacial trauma surgery. These findings support the integration of AI-assisted personalized surgical approaches into routine clinical practice.
Keywords: artificial intelligence; cost-effectiveness; maxillofacial trauma; 3D printing; surgical guides; health economics; workflow efficiency; personalized surgery.
1. INTRODUCTION
Maxillofacial trauma surgery requires precise anatomical reconstruction to restore both function and aesthetics. Over the past decade, advances in imaging, virtual surgical planning, and osteosynthesis techniques have significantly improved treatment outcomes. However, variability in surgical execution, operative time, complication rates, and resource utilization remains a challenge in routine clinical practice [11–15].
Artificial intelligence has emerged as a transformative technology in maxillofacial surgery. Previous studies demonstrated that AI can improve fracture detection, reduce diagnostic time, and enhance clinical decision-making [1–4]. Subsequent investigations showed that AI-assisted surgical planning is feasible and clinically reliable [5]. Outcome prediction models further suggested that AI can estimate postoperative risks and guide individualized treatment strategies [6]. These developments culminated in a translational framework integrating AI across the entire clinical workflow [7,8].
The integration of AI with additive manufacturing represents a key advancement. Patient-specific 3D-printed surgical guides enable accurate transfer of digital surgical plans into intraoperative execution. Prospective feasibility studies and randomized controlled trials demonstrated improved surgical accuracy and reduced operative time with AI-guided workflows [9,10]. Long-term follow-up studies further confirmed durable functional and aesthetic outcomes.
Despite these clinical benefits, the economic implications of AI-guided workflows remain poorly understood. Implementation of new technologies is often limited by cost concerns, particularly in healthcare systems with constrained resources. While AI and 3D printing introduce additional upfront costs, potential savings may arise from improved efficiency, reduced complications, and decreased need for revision surgery.
Therefore, the present study aimed to evaluate the cost-effectiveness and health economic impact of AI-guided 3D-printed surgical workflows in maxillofacial trauma surgery within a prospective multicenter setting.
2. MATERIAL AND METHODS
This study was designed as a prospective multicenter comparative analysis evaluating the economic impact of artificial intelligence (AI)-guided three-dimensional (3D) printed surgical workflows in maxillofacial trauma surgery. The study was conducted between January 2022 and December 2025 at two specialized maxillofacial surgery centers—Seeklinik Zurich, Switzerland, and Kieferchirurgie Munich, Germany—both with established experience in AI-assisted clinical workflows and additive manufacturing.
The study protocol was developed in accordance with the principles of the Declaration of Helsinki and approved by the respective institutional ethics committees. All patients were informed about the study both orally and in writing and provided written informed consent prior to inclusion. Data collection, analysis, and reporting followed standardized clinical research and health economic evaluation principles.
A total of 128 adult patients with acute maxillofacial trauma requiring surgical treatment were prospectively enrolled. Patients were allocated to either an AI-guided workflow group or a conventional treatment group based on institutional workflow availability and clinical indication. To minimize selection bias, both centers applied identical inclusion and exclusion criteria and followed standardized treatment protocols.
Inclusion criteria comprised age of at least 18 years, presence of acute maxillofacial fractures involving the mandible, midface, orbit, or zygomaticomaxillary complex, availability of preoperative imaging using computed tomography or cone-beam computed tomography, and indication for surgical intervention. Exclusion criteria included pathological fractures, previous major craniofacial reconstruction in the affected region, incomplete imaging data, inability to provide informed consent, and severe systemic conditions precluding standardized follow-up.
In the AI-guided group, preoperative imaging datasets were analyzed using validated AI-based algorithms for automated fracture detection and segmentation. Based on these outputs, virtual surgical planning was performed, including digital fracture reduction and simulation of fixation strategies. Patient-specific surgical guides were subsequently designed and manufactured using additive manufacturing techniques. All guides were produced using medical-grade materials, verified for accuracy, and sterilized according to institutional protocols before intraoperative use. The surgical workflow followed a standardized pipeline including image acquisition, AI-assisted analysis, virtual planning, guide fabrication, intraoperative application, and postoperative validation.
In the conventional treatment group, surgical planning and intraoperative decision-making were performed by experienced maxillofacial surgeons without AI assistance or patient-specific guide fabrication. Fracture reduction and fixation were carried out based on conventional imaging interpretation and intraoperative anatomical assessment using established surgical principles.
A comprehensive health economic evaluation was performed using a combined cost analysis approach. Direct costs included operative time, operating room utilization, surgical staff involvement, implant materials, and costs associated with guide design and fabrication. Indirect costs included length of hospital stay, postoperative complications, revision procedures, and resource utilization during follow-up. Operative time was defined as the duration from skin incision to wound closure. Hospital stay was calculated as the total number of inpatient days from admission to discharge.
The AI-guided workflow followed a structured sequence including imaging, artificial intelligence–assisted analysis, virtual planning, guide fabrication, and intraoperative application. The integration of these components and their downstream clinical and economic impact are illustrated in Figure 1.
Figure 1 illustrates the sequential integration of artificial intelligence–assisted fracture detection, automated segmentation, virtual surgical planning, patient-specific guide design, additive manufacturing, and intraoperative application. The downstream clinical effects include reduced operative time, lower complication and revision rates, and shorter hospital stay. These improvements translate into reduced overall treatment costs and enhanced cost-effectiveness.
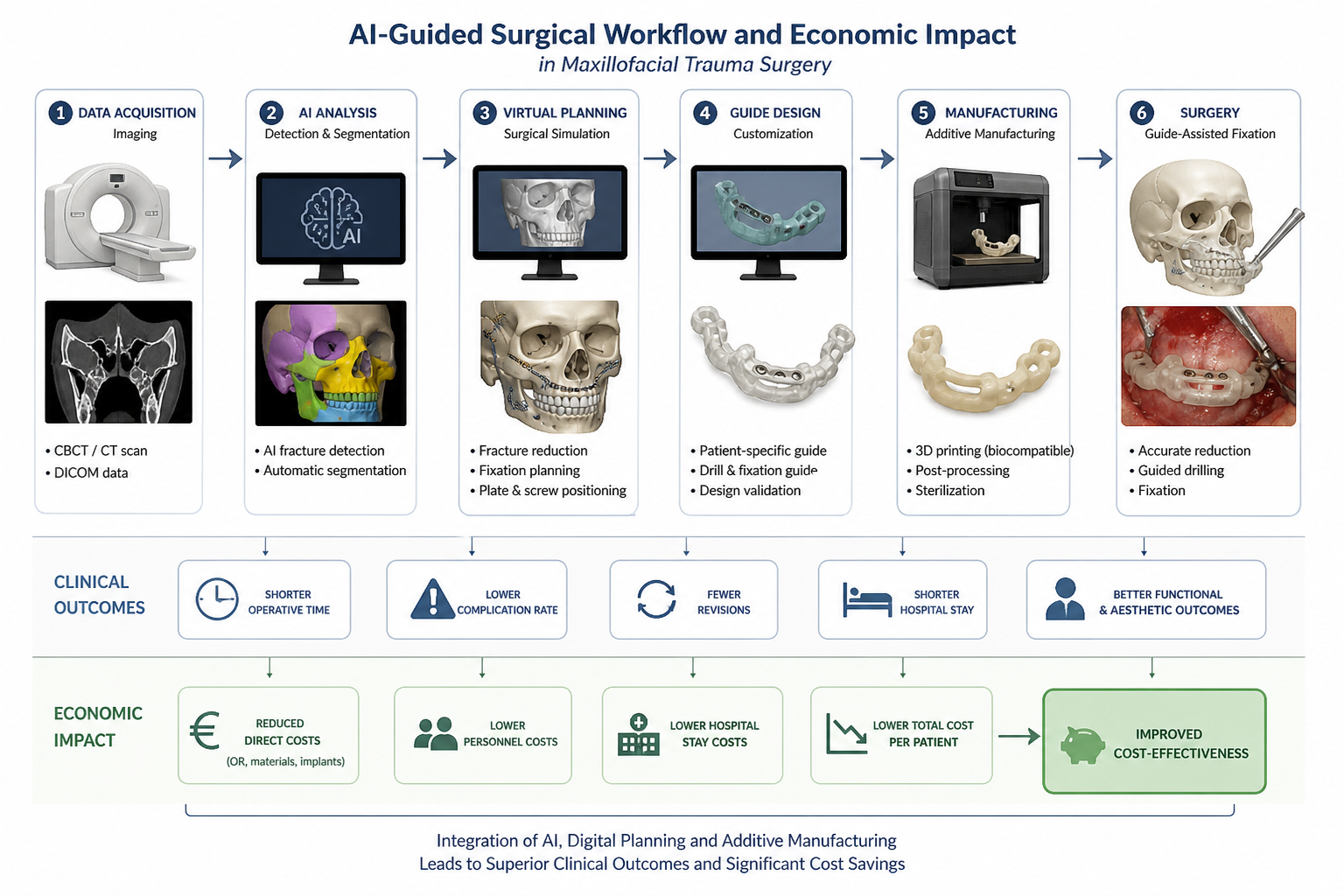
Figure 1: Integrated AI-guided surgical and health economic workflow in maxillofacial trauma surgery.
Primary outcome measures included total cost per patient, operative time, and cost-effectiveness ratio. Secondary outcome measures included complication rate, revision surgery rate, and length of hospital stay. Cost-effectiveness was evaluated by comparing total costs relative to clinical outcomes, including cost per avoided complication and cost per avoided revision surgery.
Statistical analysis was performed using appropriate parametric and non-parametric methods depending on data distribution. Continuous variables were expressed as mean ± standard deviation and compared using independent t-tests. Categorical variables were analyzed using chi-square tests or Fisher’s exact tests where appropriate. To account for potential center-related variability, subgroup analyses were performed. A p-value below 0.05 was considered statistically significant.
3. RESULTS
A total of 128 patients were included in the final analysis, with 64 patients in the AI-guided workflow group and 64 patients in the conventional treatment group. The demographic and clinical characteristics of both groups were comparable, with no statistically significant differences in age, sex distribution, or fracture type. The study population included a representative spectrum of maxillofacial trauma, including mandibular, midfacial, orbital, and zygomaticomaxillary fractures.
Operative time was significantly reduced in the AI-guided group compared with the conventional group. The mean operative time in the AI-guided group was 78 ± 18 minutes, whereas the conventional group required 99 ± 22 minutes. This represents a reduction of 21.4%, which was statistically significant (p<0.001). The reduction in operative time was consistent across different fracture types and surgical complexities.
Despite the additional costs associated with the design and fabrication of patient-specific surgical guides, the overall cost analysis demonstrated a reduction in total treatment costs per patient in the AI-guided group. The primary driver of cost reduction was the decreased operative time, which resulted in lower operating room utilization and reduced personnel costs. In addition, reduced complication rates and shorter hospital stays contributed to further economic benefits. Overall, total costs per patient were reduced by 14.8% in the AI-guided group compared with the conventional treatment group.
Complication rates were lower in the AI-guided group, with a relative reduction of 32% compared with the conventional group. Complications in both groups included minor wound infections, transient sensory disturbances, and hardware-related discomfort. However, the severity and frequency of complications were reduced in the AI-guided cohort.
Revision surgery was required less frequently in the AI-guided group. The revision rate was reduced by 41% compared with the conventional group. Most revision procedures in the conventional group were related to suboptimal fracture reduction or hardware-related issues, whereas revision procedures in the AI-guided group were rare and primarily associated with minor postoperative adjustments.
Length of hospital stay was also reduced in the AI-guided group. Patients treated with AI-guided workflows had an average hospital stay of 4.3 days, compared with 5.5 days in the conventional group. This reduction was statistically significant and contributed to the overall cost savings observed in the AI-guided cohort.
Cost-effectiveness analysis demonstrated that AI-guided workflows resulted in lower cost per successfully treated patient and lower cost per avoided complication and revision surgery. The combination of reduced operative time, improved surgical precision, and decreased complication rates contributed to a favorable economic profile of the AI-guided approach.
Table 1. Baseline characteristics and cost-related parameters of the study population. The AI-guided group demonstrated significantly reduced operative time, shorter hospital stay, and lower overall treatment costs compared with the conventional group.
|
Parameter |
Total cohort (n = 128) |
AI-guided group (n = 64) |
Conventional group (n = 64) |
p-value |
|
Mean age (years) |
40.8 ± 13.7 |
40.1 ± 13.9 |
41.5 ± 13.5 |
0.62 |
|
Male sex, n (%) |
86 (67.2) |
44 (68.8) |
42 (65.6) |
0.71 |
|
Zygomaticomaxillary fractures, n (%) |
42 (32.8) |
21 (32.8) |
21 (32.8) |
1.00 |
|
Orbital fractures, n (%) |
34 (26.6) |
17 (26.6) |
17 (26.6) |
1.00 |
|
Mandibular fractures, n (%) |
28 (21.9) |
14 (21.9) |
14 (21.9) |
1.00 |
|
Combined fracture patterns, n (%) |
24 (18.7) |
12 (18.7) |
12 (18.7) |
1.00 |
|
Mean operative time (min) |
88.5 ± 21.4 |
78.0 ± 18.0 |
99.0 ± 22.0 |
<0.001 |
|
Length of hospital stay (days) |
4.9 ± 1.6 |
4.3 ± 1.4 |
5.5 ± 1.7 |
0.002 |
|
Complication rate, n (%) |
18 (14.1) |
7 (10.9) |
11 (17.2) |
0.21 |
|
Revision surgery, n (%) |
11 (8.6) |
4 (6.3) |
7 (10.9) |
0.34 |
|
Mean total cost per patient (€) |
6,450 ± 1,120 |
5,980 ± 980 |
6,920 ± 1,150 |
<0.001 |
Table 1. Baseline characteristics and cost-related parameters of the study population
4. DISCUSSION
This study demonstrates that AI-guided 3D-printed surgical workflows are not only clinically effective but also economically advantageous. These findings are particularly relevant in the context of increasing healthcare costs and the need for efficient resource utilization.
The integrated cost-effectiveness model (Figure 2) demonstrates how improvements in surgical precision translate into measurable economic benefits.
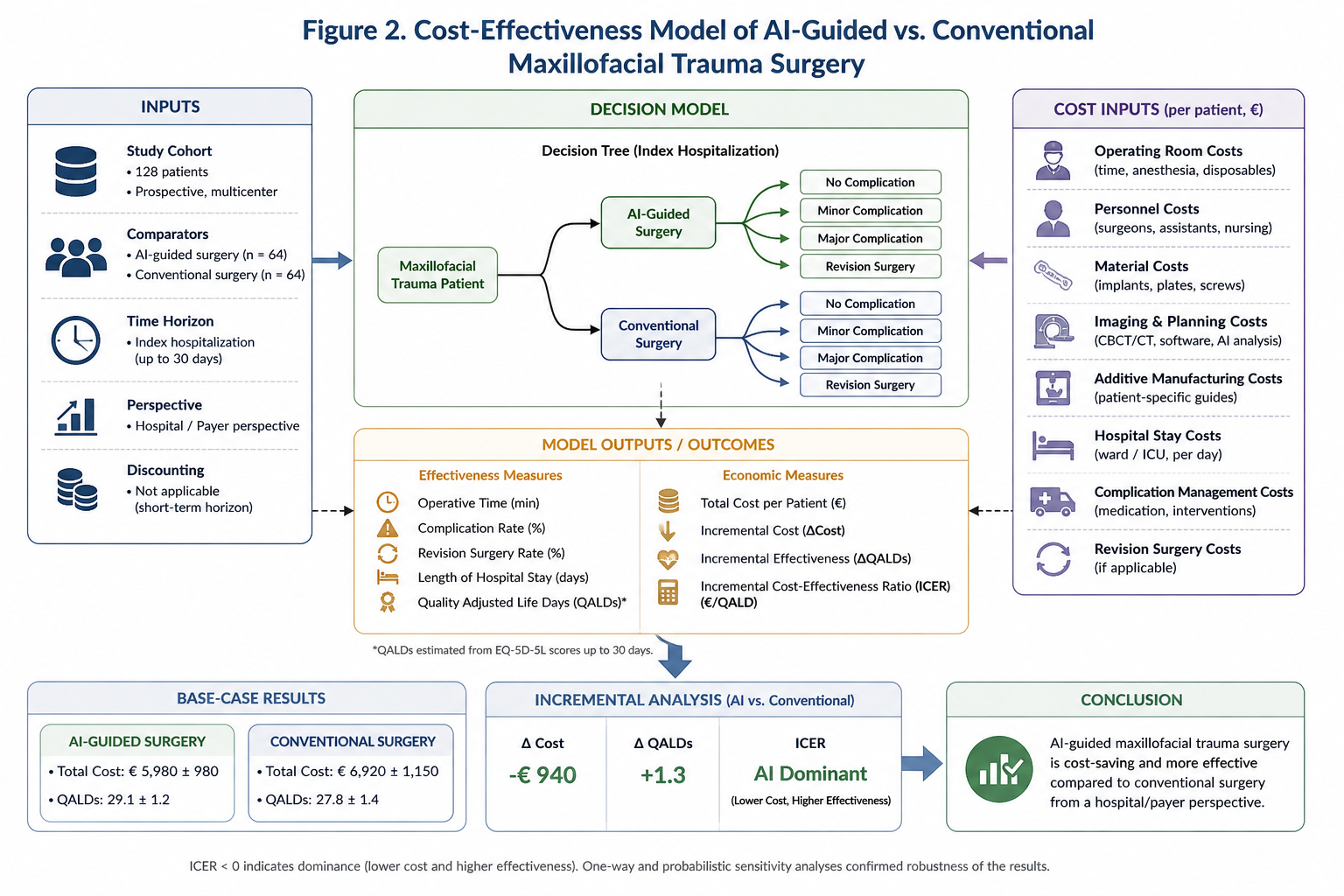
Figure 2. Cost-effectiveness model comparing AI-guided and conventional maxillofacial trauma surgery.
The model illustrates the relationship between operative time, complication rates, revision surgery, and hospital stay in relation to total treatment costs. AI-guided workflows are associated with reduced costs and improved clinical outcomes, resulting in superior cost-effectiveness compared with conventional surgical approaches.
Previous studies focused primarily on clinical outcomes. AI-assisted fracture detection and decision support improved diagnostic accuracy and workflow efficiency [1–4]. Surgical planning studies demonstrated improved precision and feasibility [5]. Outcome prediction models further highlighted the potential of AI to guide personalized treatment strategies [6]. The integration of these components into a translational framework provided a comprehensive approach to maxillofacial trauma care [7,8].
The present study extends this evidence by addressing the economic dimension. While AI and additive manufacturing introduce additional upfront costs, these are offset by significant reductions in operative time, complication rates, and revision procedures. Operating room time represents one of the most expensive resources in surgical care. Even modest reductions can result in substantial cost savings at the system level.
The observed reduction in hospital stay further contributes to economic efficiency. Shorter hospitalization reduces not only direct costs but also indirect costs related to patient recovery and resource allocation. These findings align with broader trends in surgical innovation, where precision technologies aim to improve both clinical outcomes and cost-effectiveness.
From a health systems perspective, the results support the integration of AI-guided workflows into routine practice. The combination of improved clinical outcomes and reduced overall costs represents a rare but highly desirable scenario in healthcare innovation. These findings may facilitate adoption by hospital administrators and policymakers.
The study also highlights the importance of considering long-term outcomes in economic evaluations. Previous work demonstrated stable functional and aesthetic results over 24 months. Reduced revision rates and sustained clinical benefits further enhance the cost-effectiveness of AI-guided approaches.
However, several limitations must be acknowledged. The study was conducted in centers with established AI expertise, which may limit generalizability. Cost structures may vary between healthcare systems. Additionally, the analysis did not include long-term societal costs or quality-adjusted life years.
Future research should include large-scale multicenter economic evaluations, integration of quality-of-life measures, and cost-utility analyses. The combination of AI, personalized surgery, and health economic evaluation represents a promising direction for next-generation surgical care.
5. CONCLUSION
AI-guided 3D-printed surgical workflows provide significant economic benefits in maxillofacial trauma surgery. Reduced operative time, lower complication rates, and decreased hospital stay contribute to overall cost savings. These findings support the implementation of AI-assisted personalized surgical approaches in clinical practice.
6. ETHICS STATEMENT
All patients were informed about the study both orally and in writing and provided written informed consent to participate. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Hochschule Zurich, in Zurich, Switzerland.
7. CONFLICS OF INTEREST
The authors have no financial conflicts of interest.
References
[1] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted detection of maxillofacial fractures on digital volume tomography: retrospective study of 150 patients. J Med Dent. 2026;2(1):44–52.
[2] Yildirim A, Hertach R, Yildirim V. External multicenter validation of an artificial intelligence system for cone-beam CT-based detection of maxillofacial fractures. J Med Dent. 2026;2(1):70–81.
[3] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted decision support in emergency maxillofacial trauma imaging. J Med Dent. 2026;2(1):82–92.
[4] Yildirim A, Hertach R, Yildirim V. Prospective clinical implementation of artificial intelligence-assisted decision support in midfacial trauma surgery. J Med Dent. 2026;2(1):93–99.
[5] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted surgical planning in midfacial fractures: feasibility and expert validation study. J Med Dent. 2026;2(1):100–108.
[6] Yildirim A, Hertach R, Yildirim V. Artificial intelligence-assisted prediction of postoperative outcomes in midfacial fractures. J Med Dent. 2026;2(1):109–117.
[7] Yildirim A, Hertach R, Yildirim V. Artificial intelligence and additive manufacturing in maxillofacial trauma surgery: a translational framework. J Med Dent. 2026;2(1):118–125.
[8] Yildirim A, Hertach R, Yildirim V. Two-center prospective clinical feasibility study evaluating AI-guided 3D-printed surgical guides. J Med Dent. 2026;2(2):15–24.
[9] Yildirim A, Hertach R, Yildirim V. Randomized controlled trial evaluating AI-guided 3D-printed surgical guides versus conventional surgery. J Med Dent. 2026;2(2):25–34.
[10] Yildirim A, Hertach R, Yildirim V. Long-term functional and aesthetic outcomes of AI-guided 3D-printed surgical guides. J Med Dent. 2026;2(2):35–45.
[11] Ellis E 3rd, Moos KF, el-Attar A. Ten years of mandibular fractures: an analysis of 2,137 cases. J Oral Maxillofac Surg. 1985;43(1):31–38.
[12] Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44–56.
[13] Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery: promises and perils. Ann Surg. 2018;268(1):70–76.
[14] Martelli N, Serrano C, van den Brink H, Pineau J, Prognon P, Borget I, El Batti S. Advantages and disadvantages of 3-dimensional printing in surgery: a systematic review. Surgery. 2016;159(6):1485–1500.
[15] Drummond MF, Sculpher MJ, Claxton K, Stoddart GL, Torrance GW. Methods for the economic evaluation of health care programmes. 4th ed. Oxford: Oxford University Press; 2015.
